New Science's Report on the NIH
By Matt Faherty
April 2022
Last month, Congress seeded the Advanced Research Projects Agency for Health (ARPA-H) with $1 billion. The new agency, tasked with “moonshot” biomedical research, is modeled on the Defense Advanced Research Projects Agency, or DARPA, which emphasizes nimble and ambitious projects. ARPA-H will solicit research ideas around key topics and fund the best proposals with milestone-based contracts.
The National Institutes of Health (NIH), by comparison, invests about $40 billion into biomedical research each year, but has historically been considered risk-averse. According to a report by Science, many urged the government to make ARPA-H an independent agency to avoid the bureaucratic layers of the NIH. By the end of March, though, STAT reported that ARPA-H would exist “under the auspices” of the NIH, but report directly to the U.S. Secretary of Health and Human Services, Xavier Becerra.
This strange bureaucratic outgrowth, then, is a child of appeasement: ARPA-H fundees will have access to the vast breadth of scientists and institutional knowledge embedded in the NIH, while remaining distant enough to not get tied up in the bias and entrenched interests of the agency.
To understand why an agency like ARPA-H — tasked with finding breakthroughs in Alzheimer’s, cancer and diabetes treatments — was needed in the first place, one must first understand the NIH.
In this analysis from New Science, writer Matt Faherty synthesizes dozens of interviews with current and former NIH employees and grant recipients, as well as hundreds of documents from the academic literature, to provide a comprehensive, but non-exhaustive, overview of the NIH’s operations and impact on bioscience research.
Our findings are complex and laden with opposing truths: Yes, the NIH has been the main driver of bioscience innovation for more than 80 years. Without the hundreds of billions of dollars that the NIH has allocated to U.S. scientists, the world, perhaps, would not have CRISPR gene-editing technologies or modern CAR-T therapies to treat cancer. But the NIH, like all giant bureaucracies, suffers from structural problems that hinder its efficiency and leads to considerable, wasteful spending. The agency is risk-averse and excessively funds an aging cadre of scientists, likely at the cost of losing young scientists and missing out on ambitious, transformative ideas.
Just ten U.S. universities receive 22% of the NIH’s total extramural grant budget. Still, the agency is revered; almost every scientist that we spoke with agreed its budget should be expanded. If the NIH is the fundamental driver of American bioscience, then hopefully the organization can be induced from within, or from without, to more actively seek improvements.
In this document, we break down the NIH’s history and bias, its triumphs and failures.
Thank you for reading,
The New Science Team
Introduction
In 2006, the National Institutes of Health Reform Act passed Congress. 1 This droll, political document established a Scientific Management Review Board within the NIH. That board — comprised of Dr. Anthony Fauci, a former Lockheed Martin CEO and high-ranking NIH officials — was tasked with issuing recommendations for NIH reform; a noble and useful purpose.
The board has not held a meeting since July 2015 and has written just eight reports in total, all of them between 2010 and 2015. The committee, staffed by high-ranking officials and with the power to identify flaws and encourage reforms in the NIH, has quietly gone defunct.
That should be concerning, in part, because the NIH is a behemoth institution with an annual budget of $42.9 billion in 2021 and $51.96 billion in 2022. It consists of 21 institutes, 6 centers and 300,000 current grant recipients and spends about ten times more on bioscience than the next highest-spending government agency (the European Research Council 2) and more than the entire government spending for about two-thirds of the world’s countries. It spends 20 times more on biomedical research, per year, than the Bill and Melinda Gates Foundation. 3 And yet, it does not have a functional internal board to offer feedback or propose reforms.
The NIH is the foundational engine of modern bioscience and has contributed to countless breakthroughs, even in a purely modern context — just look at the Human Genome Project and mRNA vaccines. Currently led by Lawrence Tabak, the NIH was formed in 1930 under the Ransdell Act as The National Institute of Health. It was given a single mandate: promote public health through funding internal and external research into biomedical domains.
Research activities at the NIH are still divided into two categories: intramural and extramural. Intramural research, which constitutes 10-12% of the NIH’s annual budget, is conducted in NIH labs on NIH property by NIH employees and contractors. Currently, there are over 5,000 individuals conducting intramural research, including 1,200 principal investigators and 4,000 postdoctoral fellows.4 Extramural research is carried out by universities, non-profits, hospitals, and companies with NIH funding that amounts to around 80% of its annual spending. Roughly two-thirds of this extramural spending goes directly to 300,000+ researchers5, while the remaining third goes to host institutions to reimburse their expenses related to government-funded research.
In its 90 year history, the NIH has largely staved off politicization; its inflation-adjusted budget has gone up 52% over the last 30 years. Support for government-funded research regularly polls in the high 70 to 80 percent range.
Although the NIH is a division within the larger Department of Health and Human Services (HHS), its funding is determined independently by Congress. Since 1938 (the earliest year with financial data), the NIH’s annual budget has decreased only six times.
Tens of thousands of people benefit from the NIH through employment. Hundreds of thousands of researchers at universities, private labs, and companies benefit from the NIH through grants. Hundreds of universities, including nearly all of America’s most elite universities, also benefit from the NIH through grants, a large portion of which (more than $10 billion per year) goes straight to those universities and subsidizes their facilities. Pharmaceutical and biotech companies benefit from the NIH by generating basic research, which they then turn into intellectual property and marketable products. The general public benefits from the NIH by reaping the benefits of bio-scientific advancements.
It’s not surprising, then, that few people march in the street to reduce the NIH’s budget. No politician campaigns on cutting funds for Alzheimer’s research. Private industries, non-profits and interest groups don’t have a concerted interest in fighting the NIH, which ostensibly exists to promote public health, and is ultimately answerable to the American people.
While this popularity can be seen as evidence of its success, we should also recognize the perils of the largest research organization on earth being a government bureaucracy with overwhelming support and virtually no opposition.
The NIH’s extramural research is systematically biased in favor of conservative research. This conservatism is a result of both institutional inertia, concerns by the NIH leadership that the organization could lose the support of Congress, and efforts by NIH beneficiaries to maintain the status quo.
The extramural grant distribution process, which is run through peer review “study sections,” is badly in need of reform. Though there is considerable variability among study sections, many are beset by groupthink, arbitrary evaluation factors, and political gamesmanship. The NIH may be hamstringing bioscience progress, despite the huge amount of funds it distributes, because its sheer hegemony steers the entire industry by setting standards for scientific work and priorities.
Most problematic, the NIH is highly resistant to reform. Many proposals have been shot down during discussion phases, or scaled back before implementation. The NIH’s own internal review board has been inactive since 2015, as mentioned at the start of this report section. Still, many of the NIH’s problems are likely a natural product of being a $40 billion+ per year government bureaucracy.
To understand this duality and the inherent complexity of the NIH, I interviewed 41 people and had more informal discussions with about half a dozen more while drafting this article. Eleven sources formerly or currently work in the NIH intramural programs, 24 currently or formerly received extramural funding, 18 currently or formerly served on or ran study sections, five held leadership or advisory positions at the NIH, and six held leadership positions in other bioscience research funding institutions.·
I did my best to talk to a wide range of people, spanning mainstream and heterodox positions. Some interviewees had decades of experience with NIH funding, while others left scientific research partially out of a dissatisfaction with academia.
Every person that I interviewed was granted anonymity. Despite that promise, quite a few said, during the interview, that they would be concerned about their jobs or ability to get a grant from the NIH in the future if they were publicly attached to a criticism of the NIH. One interviewee referred to a “fortress mentality” within the organization.
A few also stated that, regardless of how much I guaranteed anonymity, many researchers would refuse to talk to me because the risk was too great. One interviewee asked three other researchers to talk to me, and all three declined explicitly on these grounds.
This is concerning, in part, because the NIH undoubtedly has flaws, it is drifting away from basic research, and it is in clear need of reform.
Through my research, I have attempted to establish a comprehensive evaluation of the NIH. My objective is to both present a synthesized consensus of views on the NIH and its many components, and to present dissenting views, especially since many issues at the NIH have provoked mutually incompatible stances on how to improve operations.
My goal is to understand what works and what doesn’t work, the nature of the NIH’s incentive structure, where the organization can most be improved, and how the NIH impacts American and global bioscience.
I admit that despite the length of this work (33,000 words), I have not covered everything about the NIH. I did my best to broach every interesting topic, but based on feedback from reviewers, I know I have left plenty unsaid; especially the NIH’s impact on scientific journals and publishing. Hopefully others will build on what I have written.
Part 1: Big Picture
The NIH is Highly Regarded
I think the key issue in this large-scale assessment is quality vs. quantity. Has the NIH pushed global science forward because it has funded so many great researchers? Or, because it has so much money, is it pretty much impossible not to fund great researchers when the vast majority of that money gets pumped into elite research institutions?
With a few exceptions, all interviewees agreed that the NIH funds lots of good research and has been invaluable to global bioscience progress. Many praised the government’s role in providing funding to so many researchers for over half a century. One interviewee said he “has a tremendous love for the NIH.” Another interviewee, who was born overseas, said the NIH is “part of what makes America great.”
When asked whether the NIH’s budget should be increased, decreased, or remain the same, nearly all respondents supported increases. Most commonly, they supported a doubling or even tripling of the NIH budget. Even many of the more critical interviewees supported budget increases. I would rate only five interviewees as net-negative on the NIH as a whole, none of whom had long histories of interactions with the NIH. All emphasized that the NIH’s policies created incentives that rendered its research overly conservative (in an institutional sense), too concentrated in top-level institutions, and likely slowed bioscience research on the margin.
The NIH Is Vital to Careers
Attaining an NIH grant is nearly essential to having a career in bioscience.6 One interviewee called it the “bread-and-butter” of research, while another called it the “lifeblood” of bioscience research. Multiple interviewees noted that universities de facto require the attainment of multiple NIH or comparable federal grants to become professors and attain tenure.
This trend is driven more by economics than anything else. NIH grants are more numerous, pay higher amounts, and last longer periods of time than any other bioscience grants given by the public or private sector. Or, rather, NIH grants have the combination of all three of these qualities, whereas other top institutions only have one or two of them. For instance, there are grants that provide equivalent amounts of money and (often more) time, like from the Howard Hughes Medical Institute (HHMI), but these are far less numerous and therefore harder to get. While most extramural NIH grants have above a 20% chance of acceptance (including multiple submissions), the HHMI approves fewer than 1% of grant applications.
Thus, if a biomedical researcher was, for some reason, highly motivated to get adequate funding for a major project without NIH help, he would probably have to cobble together numerous grants from other foundations, which would likely require even more work than the NIH’s notoriously bureaucratic application process. And even if this could be accomplished, many interviewees noted it would come with significant career penalties.
Non-profit institutions pay lower indirect cost rates than the NIH, so universities tend to discourage them (more on this later), while money from private companies is considered inherently suspect within academia due to the potential conflicts of interest.
For better or worse, the NIH has a quasi-monopolistic position in the bioscience grant market. I asked numerous interviewees if this trend has a crowding-out effect (i.e. other grants have less demand or are altered in some way by the prominent position of the NIH), and most said that there was no crowding-out. Virtually all NIH grant recipients also apply for other grants, and most end up with secondary grants to support their NIH grant.
A few interviewees were strong dissenters and argued that the NIH had a “warping” effect on bioscience. One claimed that the central importance of NIH grants likely caused the protocols of other grants to adapt to NIH standards, both in an explicit structural sense, and in broader research priorities. After all, researchers have finite time, so they are likely to base their own research priorities around the aims of the largest grant recipient. This gravity well may have shaped the entire bioscience industry, again, for better and worse. Based on my own research, I’m inclined to agree with this stance, especially given the incentives created by the NIH for universities.
Because the NIH’s position in the bioscience industry is so hegemonic, it is incomparable to anything else, except maybe the European Research Council (ERC). You can’t ask, “does the NIH fund research more efficiently than the Howard Hughes Medical Institute or the European Research Council?” because the NIH spends 40X more per year than the HHMI and 10x more than the ERC. And, as one interviewee put it, “there is no Stanford in Denmark”.
Because the NIH is incomparable to any other existing organization, it’s extremely difficult, if not impossible, to accurately evaluate its efficiency level. This is crucial to understand this piece and the NIH as a whole.
Things could be, or already are, getting worse over time. Without any real competition, properly incentivized oversight or countervailing forces, no matter how inefficient it gets, the NIH will not go out of business, and is extremely unlikely to lose significant funding given the popularity of government-funded research and the support of major stakeholder beneficiaries.
The Boom Decade
From 1993-2003, the NIH’s budget increased 164%, rising from $10.3 billion to $27.2 billion.7 For comparison, in the same time frame, the Department of Defense’s budget increased by 39%,8 the Department of Agriculture’s budget increased by 17%,9 10 the Department of the Interior’s budget increased by 14%,11 12 and the entire federal budget increased by 53%.13 Even the National Science Foundation only had a 95% increase.14
I think this Boom Decade had a much larger impact on the modern NIH than its proponents at the time realized. The short-term euphoria of scientific expansion may have induced distortionary effects on the NIH and bioscience research industry, which are responsible for many of the issues outlined in this paper.
Post-Boom Decline
Roughly 80% of the NIH’s budget is spent on extramural grants given to institutions outside the government. And about 80% of that extramural spending goes towards research conducted in universities. 15
Thus, through the NIH, the federal government rapidly injected an enormous amount of cash into the university and private lab system – tens of billions of dollars over a decade with expectations of steady, if not increasing, funding in the future.
This massive infusion pushed the American research university system into an expansionary phase by incentivizing the construction of more laboratories and the hiring of more researchers and administrators to increase the capacity to earn even more NIH money. But then, in 2004, the NIH budget grew by a measly 3% and then basically flatlined in nominal terms from 2003-2015 (or shrank in real terms).
Universities and the bioscience industry had undergone too much expansionary momentum to adjust for this sudden halt in spending growth.
Thus, a mismatch in supply and demand formed. The number of bioscience researchers and labs continued to rapidly grow, just as they had done during the prior decade, but the federal research-driven demand stopped growing.
This mismatch may be responsible for many of the NIH problems outlined in this essay:
- The NIH is widely considered to be “underfunded,” despite currently being at its highest funding level ever in both nominal and real terms (albeit only recently in real terms).
- The fierce competition felt among NIH grant applicants may be a product of universities expanding their research capacity during the Boom Decade beyond what the NIH extramural budget can reasonably fund.
- As a result of that increased competition, study sections – the groups that evaluate NIH grant applications – have become more political and arbitrary.
- The rapid growth of the NIH’s budget gave it a quasi-monopolistic position in the bioscience industry, causing a gravity well effect whereby other institutions began adopting their practices and standards to the NIH.
- The universities may have gotten “addicted” to the influx of federal funds, and adapted their operations based around absorbing more federal money.
- The boom and bust may have strengthened the entrenched interests (i.e. the individuals and institutions who best capitalized on the boom) and then solidified their power since the money influx stopped suddenly after it had already been absorbed by the entrenched interests with little room for new players. This could have played out on the institutional level through universities building up huge research capacities, and at the individual level by prominent researchers amassing many large grants.
- Study sections and the NIH in general may have become more risk-averse in the dispersion of their funds now that there are so many institutional beneficiaries with entrenched interests.
Nearly all of my interviewees advocated for increasing the NIH’s budget, and many suggested drastic increases. If the 1990s and early 2000s are a good indicator, then a massive increase in the NIH’s budget could provoke the same effects again: a boom and bust cycle that, ultimately, results in unsustainable university expansion, a brutal job market for young researchers, and a less efficient NIH as an engorged bioscience research industry grows past its demand.
Entrenched Interests
Ideally, the NIH would fund research in a manner that maximizes potential long-term scientific progress within the bounds of its budget and power. But the NIH is a government-run organization and is naturally beset by political pressures that distort its structure and spending priorities.
Universities, research institutions, and major non-profit advocacy groups engage in lobbying, political pressure, and backdoor channels to push for NIH expansion, direct NIH funds toward preferable ends, and control NIH policies for their financial benefits.
Combined, these factors doubtlessly impact NIH operations and push it away from being the ideal, objective steward of taxpayer money. As far as I can tell, the NIH leadership does its best to support optimal research priorities (or at least what they perceive to be optimal), but the NIH is a government institution and is inevitably subjected to political forces.
While a few interviewees had a strong sense of this political distortion narrative and consider the NIH to be thoroughly compromised by special interests, the majority disagreed. From their point of view, although outsized benefits go to relatively few individuals and institutions, this may very well be the optimal distribution for the sake of efficiency because these entrenched interests are legitimately the best marginal researchers.
Institutional Conservatism
The single most consistent criticism of the NIH that I heard from sources, across all issues, was that the organization is too “conservative.” That is, too conservative in an institutional sense, not an ideological sense.
The NIH is considered insufficiently willing to take risks. This can be seen in its consensus-based grant evaluation, the de facto discouragement of ambitious grants, its drift away from basic research, and the lopsided distribution of grants which favor large, established organizations and researchers.
But the conservatism is most strongly felt in the NIH’s resistance to reform efforts. Throughout this essay, I’ll describe many critiques of aspects of the NIH’s operations, and I’ll describe even more proposed reforms. Yet, while reform discussions are common in and around the NIH, actual implementation of reform is vanishingly rare. The study sections, the grant protocols, the indirect cost system, and so many flawed aspects of the NIH have barely changed over the last thirty years. Most of the few reform efforts that have been implemented have failed or were scaled back, as I’ll demonstrate with the Grant Support Index and New Generation Researchers Initiative.
And, as mentioned, the NIH’s own Scientific Management Review Board, tasked with suggesting reforms and improvements, has not held a meeting or published a report since July 2015.
Francis Collins
Francis Collins was appointed director of the NIH in 2009. Before that, he led the National Human Genome Research Institute (NHGRI) within the NIH for 15 years. In December 2021, Collins stepped down from the directorship and took over a lab in the NHGRI.
With a 12 year reign, Collins is the Franklin Roosevelt of NIH directors. Most NIH directors come and go with the changing political winds, and they rarely survive new administrations. So how did Collins manage to stay in power for so long?
Seemingly, he survived by being the extremely rare individual who perfectly threads multiple needles in a niche, political realm. Namely, Collins is a progressive with a strong scientific background whose scientific viewpoints align with mainstream, left-of-center opinions on key issues, such as evolution and stem cells.
But Collins is also an outspoken, born-again Christian who literally wrote a book on how to merge science and faith. He is personally uncomfortable with abortion, but doesn’t want it outlawed.16 He is the perfect combination of the political left and right in the realm of science. A few of my interviewees talked about Collins and at least one knew him personally. Their evaluations were almost universally positive, especially on a character level. One interviewee said Collins did a “spectacular job” and saved the NIH from massive budget cuts threatened by President Trump (a 18% budget cut was proposed, but was never enacted by Congress). Others were quick to praise his hard work, charisma, and general competence.
However, there were two recurring descriptions of Collins which many would consider criticisms. First, he was often described as “conservative,” again not in an ideological sense, but in an organizational sense. Second, he was often described as more of a politician than a scientist, at least during his tenure as director.
Combined, for better and worse, the perception is that Collins took a defensive leadership posture designed to protect the NIH, and he did so quite successfully, having finally guided the NIH out of its 12 year budget slump and then stopping President Trump’s two proposed budget cuts. But to achieve these goals, Collins may have sacrificed some of the NIH’s efficiency, dynamism, and long-term potential.
One of my interviewees was a former high-ranking official of the NIH, who says they personally know Collins quite well. They praised many of Collins’s personal characteristics, but said he is “not visionary” and “doesn’t like advice.” This interviewee blames Collins for orienting many NIH policies (peer review structure, grant structures, grant types, etc.) around big institutions and translational research, and away from high-risk experimental research. They identified at least one major instance where Collins crushed an attempt by an outsider being brought into the NIH to restructure its grant system to spread funds to smaller labs. In other words, Collins either encouraged or permitted many of the biggest criticisms I heard from other interviewees, possibly as a means of currying favor from the NIH’s largest beneficiaries so they would protect the NIH.
However, this interviewee and many others noted that being the director of the NIH is an extraordinarily difficult job, which necessarily involves making compromises between multiple factions and facing constant scrutiny from opportunistic critics.
For instance, one of Collins’s biggest controversies throughout his tenure was not an increasing shift away from basic research or the failure of the Next Generation Researchers Initiative, but his approval of a $3 million grant to the University of Pittsburgh, which involved grafting fetal tissue onto mice. That sum of money is nothing to the NIH budget and I doubt Collins had any personal input into the grant approval, but nonetheless, it was a lightning rod for his career and courted a flurry of attacks from politically conservative forces. He was called a “national disgrace;"17 anti-abortion groups called for his resignation.18
Collins left office in December 2021, but his influence certainly isn’t gone. His successor, Lawrence Tabak, is a long-time lieutenant. Collins still works in the NIH, and was appointed the scientific advisor to President Joe Biden.
Part 2: Research Conservatism
A survey conducted by Fast Grants found that, if bioscience researchers had complete research freedom, 78.5% would change their current work “a lot” and 20.3% would change their current work “a little.” Only 1.2% wouldn’t change their current research.19
Furthermore, 81% of respondents said they would make their research more ambitious if they could, 62% would prefer to pursue research outside their field, and 44% said they would pursue counterintuitive hypotheses.20
Generally, I think there is good evidence that the NIH systematically promotes conservative research and is overly biased against ambitious research.
At least one interviewee, for instance, said they believed that the vast majority of NIH-funded research is worthless, at least compared to optimized, alternative research funding methods. However, the more moderate position, expressed by a vast majority of interviewees, is that current scientific progress is slower than it could be due to NIH research conservatism.
Aversion to High-Risk Research
One interviewee said that they had a graduate school professor whose final assignment was to write an R01-type grant proposal (the standard NIH grant). After everyone had turned in their projects, the professor told the class that their proposals were excellent, innovative, and he would love to see many of them come to fruition… but the NIH would never actually fund these proposals. They were too high-risk.
Interviewee estimates of how much high-risk research was funded by NIH extramural grants varied widely, from almost none, to one-third of grants.
How Does the NIH Do So Much Basic Research If It’s So Conservative?
Nearly all interviewees said that NIH research is too conservative. And while most interviewees agreed that the NIH is doing less basic research than it should, it still does lots of basic research (51% of grants). But isn’t basic research, which tends to ask fundamental questions about biology without clear answers, inherently not conservative?
So how can the NIH simultaneously do so much conservative and basic research?
The answer, as far as I can tell, is that the NIH’s structures seem to incentivize uniquely conservative, basic research.
Basically, the NIH pushes a significant portion of its extramural grant applicants into open-ended, basic research paths. But the NIH punishes researchers with high-risk, ambitious proposals within these open-ended paths. Researchers often start with moderately open-ended research goals, then secretly (i.e. outside grant boundaries) conduct experiments to get preliminary data. As the data comes in, they narrow the scope and goals of the research to a more marginal goal with clearer “deliverables.” Then, they apply for a basic research NIH grant with this narrow, marginal goal.
In practice, this means that most NIH grants, even basic research ones, are quite narrowly focused. Even if an exciting and open-ended research path has dozens of grant applicants working within it, each one is aimed at a highly-likely result, which will yield incremental advancements in the field. Open-ended, innovative projects that could either make great breakthroughs or yield null results are largely ignored by grant review panels. The most innovative projects are secretly pursued outside of grant boundaries, but are usually abandoned for safer, narrower scopes as time and funding wind down and the lab needs to apply for a fresh grant.
How is High-Risk Research Discouraged?
This is a more complicated question than it might seem. There is no high-level mandate to move the NIH away from open-ended projects. Extramural research grants are awarded by study sections whose leaders are appointed by the NIH, but whose membership consists of dozens of scientists from universities. The conservatism of NIH grant distribution can best be attributed to the structure and evaluation parameters of study sections, as well as the hegemonic position of the NIH in the bioscience research industry.
Averaging of Research Proposals
The study section process will be discussed at length in Part 4, but a crucial factor that may bias their outcomes towards conservatism is that they choose to award grants based on a consensus process determined by averaging.
Study sections generally consist of between 10 and 30 reviewers who are selected due to their expertise in a particular domain. These reviewers, in many cases, are working on research projects that are related to the very grants they are assigned to review.
Each reviewer gives a score (1 to 9, with 1 being the best) to every grant proposal, these scores are averaged, and then the average is converted into a percentile against all other grant applications in the cohort. All grants below a certain percentile (known as the “payline”) are funded.
Given the scoring system, an NIH grant application is more likely to get approved if it’s liked by everyone rather than loved by some and hated by others. Hence, researchers often design and submit projects that have broad appeal and aren’t too novel or ambitious, and which won’t lead to the invalidation of the work of anyone sitting on the review panel (which often happens as science advances). This is incrementalism: Instead of discovering something truly novel or demonstrating that some of our knowledge was wrong, approval by committees of active scientists means that researchers must build on what’s already there.
“Deliverables” Restrict Research
Many interviewees blamed the lack of high-risk research on study sections emphasizing “outcomes,” “hypothesis-driven research” or “deliverables.”
Basically, the NIH wants its extramural grants to have clear research goals with obvious practical applications. This seems to be the result of a relatively recent push, and was not as much of an issue 20+ years ago.
The demand for clear “deliverables” might seem reasonable, but it counterintuitively punishes ambitious proposals. High-risk projects tend to be more open-ended and unpredictable, and thus are heavily penalized in an evaluation process that asks researchers to enumerate specific hypotheses and likely outcomes which can be translated into practical, public health benefits. In contrast, narrow, conservative projects, with less interesting but more predictable results, are rewarded.
Some interviewees felt that this narrowing of scientific parameters ran counter to the nature of scientific progress. Discovery is just as often unpredictable or random as it is planned.
One interviewee told me an anecdote about microbiologist Dr. Thomas Brock. 21
In 1964, Brock visited Yellowstone National Park on vacation and was intrigued by the greenish-blue coloring of the hot springs. A park ranger told him that it was algae, which Brock didn’t think was possible since nothing could live in waters that hovered near 200 degrees Fahrenheit and sometimes boiled.
Brock spent six years analyzing samples from the hot springs and found that, contrary to established scientific wisdom, bacteria could survive at these high temperatures, including a microbe called Thermus aquaticus. The discovery was a paradigm shift in bioscience and microbiology.
A decade later, biochemist Kary Mullis was working on a process to rapidly amplify and copy DNA. He couldn’t find an enzyme that could tolerate extremely high temperatures, until he stumbled across Brock’s Thermus aquaticus. In 1993, Mullis was co-awarded the Nobel Prize in Chemistry for inventing the PCR process, a foundational tool for modern genetic research and treatment development, and more recently used as a primary method for COVID-19 testing.
Note that Brock’s random discovery enabled an entirely unexpected and unrelated major advance in science. First, a scientist randomly stumbled upon something interesting while on vacation, ushering in a paradigm shift in the scientific understanding of the limits and adaptability of life. That breakthrough unexpectedly identified the perfect component to insert into a process that rapidly manufactures DNA.
This is not to say that the NIH should fund any random curiosity that pops into a researcher’s mind, nor that the NIH should ignore broader scientific objectives (like curing particular diseases). But many researchers believe that the NIH’s structure hinders genuine curiosity-driven research, which often starts with tangential questions and end with broadly useful results.
Preliminary Data, Unspoken Requisite for Funding
Another much-maligned promoter of conservatism is an emphasis on preliminary data. Study sections want to see pre-existing work, either from the applying researcher or other sources, which demonstrate the viability of their hypothesis. High-risk projects are far less likely to have pre-existing data because, if they are truly novel, no one will have done anything like them before.
However, after hearing complaints about preliminary data requirements by maybe a dozen interviewees, I was surprised to learn that it is not a requirement at all, at least not officially. That is, the NIH study section criteria does not officially require preliminary data. Rather, having preliminary data is basically an informal norm for current grant evaluation standards.
Study Section Leadership
Some interviewees suggested that the NIH’s managers are pushing for more conservative research through the purposeful selection of the study section leaders, or Senior Research Officers (SROs). Whether by design or accident, the NIH has supposedly been choosing more conservative SROs, who in turn pick conservative study section reviewers, who then select conservative grants. Study section leaders tend to be older and more well-established in their fields; there may be some selection bias at play.
Most Grants Propose “Safe” Aims
Finally, grant applications are supposed to have clear goals, and if those goals aren’t met, the grantee is unlikely to get funding in the future. In the words of one interviewee, “if you make a big leap and you miss, you’re done for.”
This plays into recent calls across the scientific community for more tolerance for research that yields null results (see [Nature]* and [Inside Science]*). Scientists make empirical observations, devise a hypothesis about a causal explanation for the observation, and then design experiments to isolate variables that determine whether a single variable is causal; but there is no default reason to expect that the hypothesis is true. And the more ambitious and speculative the research, the less likely the hypothesis is to be true.
Pushback Against Claims of NIH Conservatism
Upon showing this subsection to researchers, some pushed back against my argument. They did so not because they thought NIH-funded research was sufficiently ambitious, but because they questioned whether such a broad and substantial assessment could be made about the tens of thousands of grants given out by the NIH each year, and based upon the approval of thousands of reviewers, by interviewing 41 individuals and scouring journal articles.
That’s a fair critique. I couldn’t find any good studies comparing the ambitiousness of NIH-funded research to other institutions, except in narrow cases, like how the NIH’s handful of specially designed high-risk grants stack up against Howard Hughes Medical Institute researchers. Indeed, the size of the NIH makes good comparisons impossible, since all other organizations can afford to be more selective.
There’s also the lingering caveat that interviewee testimonies could represent biased opinions. Many of my interviewees relayed personal stories of grants they considered excellent (usually their own) getting shot down, and it’s possible that they just had a difference of opinion from more sober minds.
But I still think that the NIH extramural research program is biased in favor of conservative research.
For one, this opinion was held by almost every interviewee I asked, albeit to varying degrees. These interviewees vastly ranged in terms of age and career level. Postdoctoral fellows still learning about the system, as well as established professors with decades of NIH-funded research projects, agreed on this point.
Second, I think the structure of the study sections and their parameters create clear incentives that lead to conservatism. These incentives could be counteracted by other stronger incentives, but as will be elaborated upon more in Part 4, I don’t think these incentives are strong enough.
Third, as I’ll explain in the rest of Part 2, I think there are strong incentives for the NIH leadership to encourage this conservatism, both given the unique incentives of the NIH, and due to the default incentives of any large bureaucracy.
The “Benevolent Ponzi Scheme”
Many NIH-funded labs follow a form of the “Benevolent Ponzi Scheme” strategy. Two young graduate student interviewees said the system was already explained to them by superiors, while nearly all older interviewees confirmed familiarity with some form of it. Here’s how it works:
When a researcher becomes a faculty member and gets his own lab and staff, they are typically given a funding package of a few million dollars by the university to kick off new research. The researcher will come up with a project. Then, they will use their starter funds to complete “80-90%” of the project. Then they will apply for an NIH grant for the project, even though it’s 80-90% done.
Once the grant is in-hand, they will use a small portion of the funds to complete the project.
Then, they will use the bulk of the funds to get started on the next project. When that project is 80-90% done, they will apply for an NIH grant for that project. Then they will finish that project, and use the bulk of the funds to get started on the next project. And so on.
Why Do Researchers Use This Scheme?
The Benevolent Ponzi Scheme optimizes both grant acceptance rates and research freedom within the current NIH system.
Two of the most important criteria for grant approval are to have clear “deliverables” and pre-existing data. The easiest way to get both of those is to basically do most of the project before it’s approved. A fresh, bold, novel, high-risk project will have uncertain endpoints and no preliminary data. A project that is 80-90% done will have plenty of preliminary data and an obvious hypothesis that the data is pointing toward. Plus, since the researcher has already been working on the project for a while, they probably had plenty of time to think about applications for whatever result they have already pre-guaranteed.
That might sound utterly cynical, but there’s a more optimistic upside. Since the Benevolent Ponzi Scheme researcher can finish their official project proposal quickly and at little cost, they are free to pursue more interesting, higher-risk research with the leftover funds and time. If they make genuine discoveries, they can just claim that they decided to change research directions on grant maintenance reports. If they don’t make discoveries, then they can just turn in their original project.
Do the Study Sections Know About This?
I’m not sure, as I’ve gotten different answers from interviewees.
Some interviewees say that everyone knows about the Benevolent Ponzi Scheme. Any decent study section reviewer will assume that the grant applications they are reviewing is at least partially based on lies or omissions. Maybe the grant applicants will have conducted more experiments than they are letting on, or maybe they are pretending their hypotheses aren’t, for the most part, already confirmed by the data. It doesn’t really matter.
But some interviewees say that the Benevolent Ponzi scheme is kind of a trade secret and plenty of researchers don’t figure it out until later in their careers. Other researchers, they say, don’t end up using this strategy because they’re too confident, competent, or honest.
Could The NIH be Slowing Research? The Gravity Well Effect
A few interviewees suggested that the NIH was slowing the entire bioscience research industry by acting as a gravity well and spreading conservatism. Here’s how this could work.
If the NIH tends to fund conservative researchers, and NIH grants are the basis of the entire American bioscience research industry, then research institutions are strongly incentivized to hire conservative researchers. These conservative researchers then teach younger researchers conservative techniques, and move into the NIH study sections where they will judge grant applicants by conservative standards. And so on.
One interviewee says this incentive structure “has corrupted everything” – young researchers, career orientations, the hiring process, the universities, and even academic journals. For young researchers in particular, they claimed it was causing a brain drain, whereby the most ambitious researchers were fleeing academia and basic research in favor of biotech and pharmaceutical companies, leaving more conservative researchers behind for the remnants of basic research and the bulk of NIH funds.
How Much Real Research Freedom Is There?
A researcher’s freedom to pursue a broad range of hypotheses is stronger than their grants would suggest, because many researchers lie about their true grant parameters. Still, many people don’t have that much freedom because they need some level of fidelity on their grant applications and they need to report research progress to the NIH. How well they balance these factors is up to their grant application and management skills, which in turn is dependent upon their training.
One interviewee suggested that the more NIH money a researcher gets, the less control they have over their research. That is, with more money from their university (starter funds) and private foundations, they will tend to have more latitude in their research. But the more they rely on NIH funds, the more they will be tethered to their grant applications and updates to grant administrators.
I would think that well-established researchers would have more latitude, but even that has its limits. One interviewee related stories of two instances when their grants were rejected because they involved technology not ongoing in their lab and, thus, there was no preliminary data. In the latter case, the interviewee had been receiving NIH grants for over forty years, they had served as an editor on a major journal, and had been an advisor for an NIH institute. All that clout and history wasn’t enough to get the grant approved. While such ability to withstand political forces is impressive, the reason the grant wasn’t approved was that the interviewee never worked in the field in which they applied for the grant.
Fortunately, the proposal later caught the attention of a prominent non-profit. The interviewee submitted a one-page application and they “nearly fell off [their] chair” a few months later when they got full approval at a higher funding amount than expected. Their project has since yielded “transformational” progress in the field, and though the interviewee is extremely positive about the NIH overall, they are concerned about the lack of risk-tolerance in study sections.
Another interviewee said they once applied for a grant that involved mouse studies. The grant wasn’t approved. On their feedback forms, they found that they lost a lot of points because they didn’t yet have possession of the mice. “Well of course I didn’t… I wasn’t funded yet.”
A few researchers had an interesting take on an unintended consequence of this system: the NIH is biased against “super nerds.”
Navigating the “benevolent ponzi scheme” requires anticipating the judgments of colleagues, knowing the right people to talk to for advice, plotting out how to stagger grant timing and explain results that diverge from official grant applications. These are all skills correlated with extraversion, networking, and sociability. They are not the typical traits of a socially awkward scientist who loves to spend hours going through data sets and discussing abstract theories, rather than figuring out how to game complex bureaucratic systems.
This is not to say that a researcher can’t be both a great scientist and a skilled player of the game. But there are certainly researchers who are uncomfortable with the system, and who wish they could spend more of their time on the science and less on figuring out how to get to do the science.
On the other hand, there are some researchers who are probably a bit too comfortable with complexities embedded in the grant system. Whether by design or happenstance, some lab leaders gain reputations at being so good at getting grants that they focus most of their energy on getting resources and then leave the actual science to their staff. Then again, maybe a bunch of super nerds working for a master grant-getter is the ideal lab structure?
The NIH Does Fund Some High-Risk Grants
Despite all of the above, the NIH has a few grant programs specifically designed for high-risk research, all of which garnered praise from interviewees.
The Pioneer Award is arguably the single-most prestigious grant at the NIH. It is open to applicants from all career stages and requires no preliminary data, and it must be for an entirely novel, scientific proposal. Grants are for at least $700,000 per year for five years. I can’t find an acceptance rate, but the NIH aims to give only seven per year.22
According to the NIH’s own research (from 2013), 23 Pioneer Awards produce more citations, more publications in high quality journals, and are more innovative per-dollar than the standard R01 grants. Somewhat comparable Howard Hughes Medical Institute researchers generate more publications in top journals, but otherwise produce similar results, at least within the smaller timeframe of the Pioneer Awards.
The New Innovator Award is basically a smaller version of the Pioneer Award, but for new researchers who just completed their terminal degree. Funding is $1.5 million split across “two multi-year segments,” and the NIH gives out about 35 per year.24
The Early Independence Award is for current or recently graduated doctoral students (a group that receives vanishingly little funding through direct grants from the NIH). The NIH aims to give out 10 awards worth $250,000 per year for up to five years.25
Another interesting grant is the R35, which is structured similarly to the Howard Hughes Medical Institute’s model of funding “people over projects.” The grant requires no specific aims, but provides a steady (albeit relatively low) level of funding for eight years.
The Transformative Research Award is for “inherently risky and untested” projects, and is more oriented to big teams. In 2022, the NIH will dole out seven awards, worth a total of $8 million.26
Many interviewees said that these grants are underfunded. Even former NIH Director, Francis Collins, has advocated for their expansion.27 Going by the NIH’s guideline numbers, the organization will spend only tens of millions per year on new grants in these categories. The total extramural budget, recall, surpassed $30 billion last year.
A grant-giving non-profit, Open Philanthropy (OP), has an interesting take on the Transformative Research Award. In 2017, OP worked with the NIH on their Second Chance program. They invited researchers who had been rejected from the NIH’s Transformative Research Award to reapply for OP funding. For non-profits, piggybacking off the NIH’s infrastructure seems like a no-brainer.
OP received 120 proposals in three weeks, and funded four grants for a total of $10.8 million.
OP found “little correlation between our evaluations of the 120 proposals and the NIH peer review panel’s evaluation of these proposals.” They suggested this might be because they “[look] to support projects that could have the largest humanitarian impact, regardless of whether they ‘overturn fundamental paradigms,’ as the NIH phrases it.”
However, OP also said: “we considered many of the submitted proposals to be a bit on the conventional side. This surprised us given the ‘transformative’ premise and focus of the TRA program. We speculate that this may be due to the constraints within which applicants feel they must work to get through panel reviews.”
Basic Vs. Applied Research
In a 2017 Congressional testimony, then-NIH Director Francis Collins said:28
“The core of our mission remains basic biomedical science. Given the exploratory and, hence, unpredictable nature of fundamental discovery, basic science is generally not supported in the private sector – but it provides the critical foundation for advances in disease diagnosis, treatment, and prevention through future clinical applications.”
The consensus among interviewees, particularly those who had worked with the NIH for more than two decades, is that the organization has shifted away from basic research and more toward applied research. Admittedly, the line between these concepts can be fuzzy. Nevertheless, there is a pervasive sense that there has been a struggle between these two research categories and that the latter is winning out.
The NIH currently considers about 51% of its research to be oriented toward basic science;29 based on their own categorization, basic research has slightly declined from 56% of grants in 2003.30
Basic research is the foundation of science; its insights are far less predetermined, but have far more potential value in downstream applications. Typically, applied science is considered the domain of private industry, where medicines, treatments, and biotech can be packaged and sold for profit. The NIH and National Science Foundation favor basic science precisely because private industry allegedly lacks the incentives to do so.
The NIH’s applied research aims have expanded far beyond the gaps left unfilled by private industry, though, and the agency is now funding research into treatments for a slew of major diseases. My speculation is that this trend started with President Richard Nixon’s “War on Cancer” and the 1971 National Cancer Act, which massively expanded the NIH’s National Cancer Institute (NCI) and cemented its status as the highest-funded institute in the agency.
While most of the interviewees lamented this trend, some were positive about it. Some interviewees asserted that the NIH has done too much research in the past to fill up academic journals, without enough practical results.
Ultimately, the NIH was never intended to fund solely basic research, like the NSF. And, given the enormous scope of the NIH’s work, I’m sure there are some domains where it could be more productive with applied science. But overall, most researchers were concerned that the NIH is drifting away from its core function.
Translational Research
The drift away from basic research largely manifests as a new emphasis on “translational” research, or research that translates basic research into applied research. One interviewee summarized the support for translational research with something he overheard in a study section: “It’s the National Institutes of Health, not the national institutes of cool tricks you can do with genetic mouse models.”
There is a concerted effort to push translational research both within and outside the NIH. One interviewee argued that the NIH has produced a significant amount of useful work which has yet to yield real-world results because of a lack of translational efforts either by the government or private industry.
Why Is the NIH Moving Away from Basic and High-Risk Research?
The best explanation I heard was from an interviewee who said the NIH’s overarching goal is to attain “slow and steady progress on all fronts.”
The NIH’s budget is determined by annual appropriations doled out by Congress, and thus the NIH is highly conscious of how its operations are perceived by the public. Whether it is optimal for the NIH from a public health perspective to pursue more basic or applied research isn’t especially relevant here. What is relevant is how the public perceives these methods. If anything, there is a bias in the general public in favor of applied research because it’s more tangible.
The public is not an expert on scientific matters, and thus it is far easier for the NIH to maintain and increase its budget by promoting itself as an agency devoted to directly solving major health problems through applied research, rather than by laying the foundations for other organizations to create treatments for these health problems through NIH-funded basic science research.
This trend has only intensified in the modern era as the NIH has come under attack by a faction of conservative Republicans who accuse it of exacerbating big government waste. Republican Senator Rand Paul publishes a “Waste Report” where he often identifies NIH projects of allegedly dubious value. In 2019, he singled out an $874,000 project to see how cocaine impacts the sex drives of Japanese quails, and a $1.2 million joint study between the NIH and NSF to study online dating habits (the NSF was also targeted for a $467,000 study on Panamanian frog mating calls).31 He has also attacked a $500,000 NIH study on how much people like spicy food,32 and a $380,000 study on why college freshman tend to gain weight.33
Senator Paul’s tactics are arguably nothing more than cheap political jabs. As the Washington Post pointed out, Senator Paul got his figures wrong on the cocaine quail sex study (it only cost $357,000), and the study was an attempt to better understand how cocaine impacts sexual behavior for the sake of drug addiction treatment (the quails were used because of their highly consistent mating patterns).34
I don’t know enough about Japanese quails or the impact of cocaine on sex patterns to judge the study, but neither does almost anyone else. That’s the point. Maybe it was just scientists pulling a fast one on the agency by studying something completely meaningless and pretending that it could be useful for our understanding of drug addiction. Or, maybe it was really done because of its direct application to the problem. The sheer absurdity of the study, from the layman’s perspective, makes it an effective punchline for criticisms of government waste. Instead of spending more money on educating children or curing cancer, the government is buying cocaine for birds and watching them have sex.
I can’t say for sure that Senator Paul’s criticisms or anyone else’s are what’s pushing the NIH away from basic research, but the sentiment of numerous interviewees is that it’s having an impact. I personally don’t see any substantial, organized opposition to the NIH, so if this is triggering the “fortress mentality,” I think the NIH leadership and its benefactors are being overly defensive.
Part 3: Research Funding Inequality
The NIH was allocated $41.6 billion in 2020, about $30 billion of which was awarded to 56,000 grants. The NIH’s grant distribution approach has generally been criticized within academic articles because of its unequal distribution. Inequality in grant distribution has been on the rise since 1985, according to a 2017 article used as a citation throughout this section, and continues to rise to new extremes.35 I’ll tackle a few of the biggest examples of funding inequality and then discuss whether the current distribution is fair and/or efficient.
Researchers
In 2017, 1% of NIH extramural grantees received 11% of the total funds. The top 10% of researchers received over 40% of the funds.36 37 This is a larger divide in wealth inequality than for the general U.S. population, where the top 10% of earners received 40% of (post-tax) income.
The proximate mechanisms of this imbalance include established researchers getting larger grants, getting more simultaneous grants, and the higher likelihood of renewing grants compared to attaining a grant for the first time. However, many researchers worry that the current structure permits a snowballing effect, where established researchers amass too many grants that pull finite money from the common grant fund at the expense of newer researchers.
Institutions
The NIH gives about 50% of all extramural grant money to 2% of applying organizations, most of which are universities or research facilities attached to universities. 38 The top 10 NIH recipients (out of 2,632 institutions) 39 received $6.5 billion in 2020. This is 22% of the NIH’s total, extramural grant budget ($29.5 billion40), and 16% of the NIH’s entire budget.
In 2020, the top ten largest recipients of NIH money were: 41
- Johns Hopkins University - $807 million through 1,452 awards
- Fred Hutchinson Cancer Research Center - $758 million through 301 awards
- University of California San Francisco - $686 million through 1,388 awards
- University of California Los Angeles - $673 million through 884 awards
- University of Michigan Ann Arbor - $642 million through 1,326 awards
- Duke University - $607 million through 931 awards
- University of Pennsylvania - $594 million through 1,267 awards
- University of Pittsburgh at Pittsburgh - $570 million through 1,158 awards
- Stanford University - $561 million through 1,084 awards
- Columbia University Health Sciences - $559 million through 1,003 awards
Add up the top 30 recipients, and the sum is $14 billion. That’s 44% of the NIH’s extramural budget, and 36% of the NIH’s total budget.
These figures are even higher if you count university windfalls across multiple institutions. For instance, Harvard University is connected to Massachusetts General Hospital, Boston Children’s Hospital, and the Broad Institute of MIT and Harvard, the 12th, 42nd and 45th largest recipients of NIH funds.
The top institutional beneficiaries of NIH funding have also remained remarkably consistent over time. Of the top 50 beneficiaries in 1992, 44 remained in the top 50 in 2003, and 40 remained in 2020. 39 Of the top 15 beneficiaries in 1992, 14 remained in the top 15 in 2003, and 13 remained in 2020.
On the other hand, most NIH institutional beneficiaries get very little funding. Out of the 2,632 beneficiaries in 2020, 1,055 (40%) received less than $1 million.
Geography
The geographic distribution of extramural grants almost entirely stems from the distribution of universities throughout the country. The NIH gives about 50% of extramural grant money to just five states:42 California, New York, Massachusetts, Maryland, and North Carolina.
The five states with the least funding are, in order from least to most: Wyoming, Alaska, Idaho, South Dakota, and North Dakota. From the NIH’s most recent data, California currently receives $6.7 billion in active grants. Wyoming, $16.5 million.43
Gini Coefficients
Though grant applicants (supposedly) don’t know who their primary reviewers are, reviewers know whose grants they are evaluating. Thus, it’s possible that grant applicants from elite research institutions get a passive evaluation boost by sheer association and prestige, though this boost could be interpreted rationally (the best researchers are probably at the best universities) or irrationally (arbitrary prejudgment). 44 Small experiments in which reviewers are blinded to race, for example, show unbalanced effects on overall grant scores, and reviewers are often still able to correctly guess who wrote the grant. 45
The Gini coefficient is the standard economic metric used for calculating income inequality. Countries with less inequality, like Iceland and Slovakia, have lower Gini coefficients (typically in the 0.25-0.30 range). Highly unequal countries, like South Africa and Brazil, are in the 0.50-0.60+ range. The United States has a Gini coefficient of 0.41 and the United Kingdom, 0.35.
In 2020, the Gini coefficient for the NIH’s extramural institutional recipients was 0.47. If that were a country, it would be the 23^rd^ most unequal in the world, just ahead of Venezuela. 46
Did the Boom Decade cause a concentration of funding among top NIH institutional recipients?
Between 1993 and 2003, the NIH’s budget increased by 164%, rising from $10.3 to $27.2 billion.7 This marked shift slightly decreased funding inequality, as measured by Gini scores.
During that ten year period, earnings for the top 50 NIH recipients increased from $4.3 billion to $7.5 billion. Meanwhile, the share of extramural funding (excluding contracts) earned by the top 50 NIH recipients decreased from 59% to 55%, and the number of institutional recipients increased from 1,653 to 2,350, or 42%. The Gini coefficient slightly decreased, from 0.51 to 0.49.
Out of curiosity, I checked how the numbers on the lowest-earning NIH institutional recipients and how they fared over the Boom Decade. I picked $1 million in 2003 as a fairly arbitrary cut off point; it represents two to four R01 grants.
Of the 2,755 institutional recipients in 2003, 2,017 (73%) earned less than $1 million. In 1992, out of 1,653 recipient institutions, 1,176 (71%) earned less than $762,500 (=$1 million 2003 USD).
In other words, the distribution of NIH funds during the Boom Decade was remarkably consistent. And that fund distribution trend line remained remarkably consistent for the two decades after the Boom Decade. The percentage of extramural funding received by the top 50 beneficiaries:
-
1992 – 59%
-
2000 – 56%
-
2003 – 55%
-
2010 – 56%
-
2020 – 57%
Grant distributions are determined by thousands of scientists across hundreds of study sections and specialized domains. It’s a mystery, then, how all these people, with seemingly no coordination, distributed funds in almost exactly the same pattern over thirty years.
The Case for Current Efficiency
Whether the current, lopsided distribution of extramural grants is efficiency-promoting or dragging seems to be one of the most contentious issues in the NIH.
Defenders of the status quo have a simple but valid argument: The best researchers tend to congregate at the best institutions, they say, and so it’s reasonable that Johns Hopkins, UPenn, the top UCs, Yale, Harvard, and so forth would have the best labs, the best equipment, the best faculty, and therefore would earn the most grant money. In turn, the NIH should be funneling a highly disproportionate amount of money to these institutions for the sake of efficiency.
One of the major reasons some individuals accumulate lots of grant money is that they need multiple concurrent, consecutive grants to finance expensive research. There is indeed a snowballing effect, but perhaps it is more efficient to pile a relatively large amount of money on a relatively small number of researchers, rather than leave top-level researchers with fewer grants and less money for the sake of distributing funds to marginally worse researchers.
It’s not necessarily true, though, that the best researchers work at the highest-ranked schools. Studies show 47 that papers authored by researchers at a top school, like Harvard or Stanford, are more likely to receive a higher citation count, regardless of the quality of the work. It’s difficult to measure truly transformative research impacts.
Other studies indicate that the marginal value of NIH funding declines past a point that isn’t far from the median grant (I detail them in the next section), and thus status quo critics endorse spending caps. But these studies are simplifying complex outcomes.
Research impact often isn’t felt for many years after projects are completed, and it can’t be easily captured with blunt proxies like publications and citations. Caps might pull money away from some high-spending, low-efficiency researchers, but they will also defund the best and brightest at the NIH who have earned their huge grants by proving their ability.
Some interviewees claim that the NIH has already sacrificed efficiency for the sake of grant distribution. In particular, some said that the NIH is biased in favor of giving grants to institutions based in low population states, likely due to political concerns, or what one interviewee called an “egalitarian impulse.” If that’s the case, then critics have a legitimate qualm that the NIH is sacrificing some degree of efficiency by rewarding inferior researchers for political purposes.
The Case Against Current Efficiency
All of the above may be true, and yet grants are currently too lopsided in their distribution to optimize efficiency. I personally lean toward this opinion, albeit with a low confidence.
There are numerous studies that attempt to determine the marginal value of grants at different funding levels, but they all rely on citations and publications as metrics, which I think don’t work as proxies for quality. Not only are citations and publications highly variable in their quality, but larger, more established labs and researchers may very well publish less often and get fewer citations because they aren’t as bound to the “publish or perish” mindset of much of academia.
Nevertheless, I think there is likely somewhat of a detrimental bias in favor of top universities and established researchers simply due to the incentives at play. The entrenched interests are heavily dependent upon the NIH and have the means to influence its grant distribution process, both through official channels (as will be explored in Part 7) and through passive cultural norms which reinforce the superiority of established labs and researchers. But again, there is no strong empirical evidence to support this claim.
Proposed Reforms to Funding Inequalities
There have been calls for the NIH to cap funding per researcher since at least 1985.48 In 2017, University of North Carolina professor, Mark Peifer, publicly called on the NIH to impose a cap on funding per grant recipient. His paper suggests a $1 million cap as a soft target, which would still permit many large grant recipients, but prohibit significant, low-margin spending.49 The NIH’s grant database identifies only 542 grants made in 2020 with sizes between $1 and 10 million.50 A separate analysis of 2015 data placed a theoretical, $800,000 cap on individual researchers; that strategy would have freed up $4.22 billion (after bumping all <$200,000 grants up to $200,000) to be distributed to other researchers for higher yield projects. If the new researchers were each given awards of $400,000, then 10,542 new researchers could have been funded, constituting a 20% increase in total grants.
Even if the base data are valid, I assume there are highly talented researchers who can use higher dollar amounts effectively. Likewise, I’m sure there are projects which would benefit from a grant of, say, $450,000 rather than $400,000. Perhaps strict caps risk squeezing researchers into needless restraints, which could hurt research for the sake of meager savings.
The Failure of the Grant Support Index
Perhaps the most blatant example of a failed reform effort at the NIH is the aborted Grant Support Index (GSI).
In May 2017, the NIH announced the implementation of the GSI to limit grants to top researchers so that funds could be distributed more widely. The idea was simple: assign points to researchers based not only on the size of their grants, but also on their specific field, thereby accounting for variability in funds required for different types of research. Then, there would be a point cap (roughly equivalent to three concurrent R01 grants) which, if surpassed, would require the applicant to jump through extra hoops in future grant application processes and face lower odds of acceptance. It was by no means a draconian limit, as it would only impact 6% of NIH-funded researchers. But it was a concrete step in limiting funding concentration.51
The plan “caused an uproar among many scientists,” according to Science. Some researchers expressed legitimate grievances, like how the GSI would discourage lab collaboration, while others almost certainly just feared they would lose out on NIH dollars. Under pressure, the NIH relaxed the point requirement so that it would only impact an estimated 3% of NIH grant recipients.52
Just over one month after its introduction, the NIH abandoned the GSI entirely.
According to one interviewee, who used to hold a high-ranking advisory position in the NIH, pressure from top researchers and labs pushed then-director Collins to end the GSI. It’s impossible to prove this, but a 2017 paper corroborates the idea: 53
“Not too surprisingly, there was pushback [against the GSI], the most strident and well-publicized of which seemed to be from a small number of very well-funded scientists who seem unwilling to relinquish their hold on a disproportionate amount of NIH funds. Some of their rhetoric was heated—one was quoted in the Boston Globe as saying, ‘If you have a sports team, you want Tom Brady on the field every time. You don’t want the second string or the third string.'” 54
It seems that well-funded and powerful scientists, threatened by this new approach, tipped the balance. The Advisory Committee that made the decision to reverse the GSI did not represent the diversity of career stages affected by this critical decision, according to one interviewee.
“The reversal of the GSI policy sent a demoralizing message to many of us,” Peifer wrote. 53 “I think if you ask your junior colleagues, whose voices were largely not taken into account in this discussion, you’ll find that the vast majority of them support some sort of funding limitations. My recent conversations with colleagues suggest a significant number of senior scientists also share these concerns. The almost 1500 people who have already signed a petition to NIH Director Francis Collins to reinstate a funding cap provide an indication of the breadth of this opinion.”
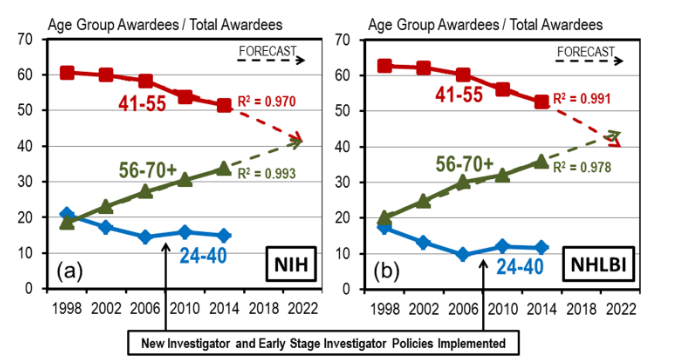
Researcher Age
The age of NIH-funded researchers has been a contentious issue for decades and is arguably one of the most apt examples of its institutional conservatism. Inequities in the age of NIH grant recipients may hinder the careers of young scientists and scientific progress as a whole.
From 1995 to 2014, R01 grantees over age 55 garnered an increase in grants of $2.5 billion, while grantees under 56 only yielded a $350 million increase. In 2014, 5% of grantees were over 71, compared to 1% in 1995.
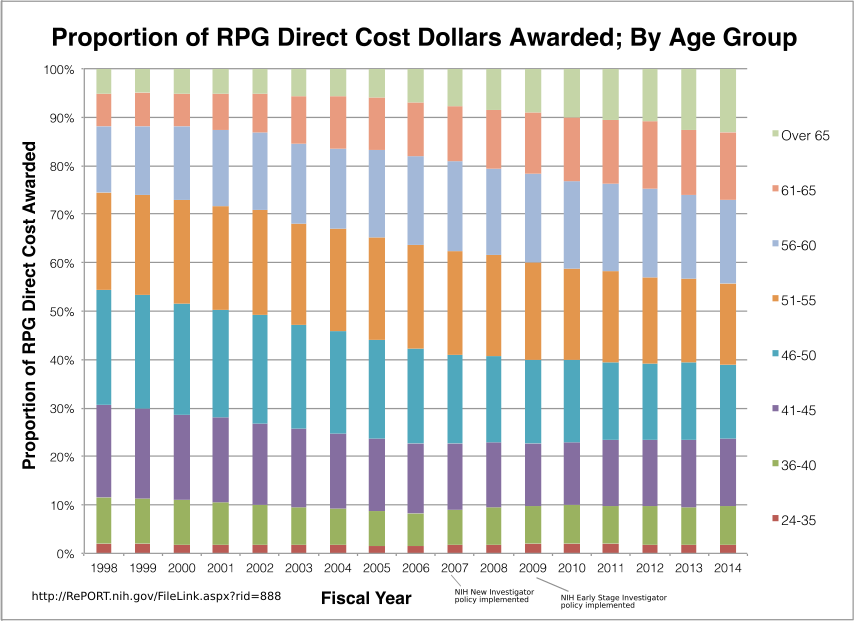
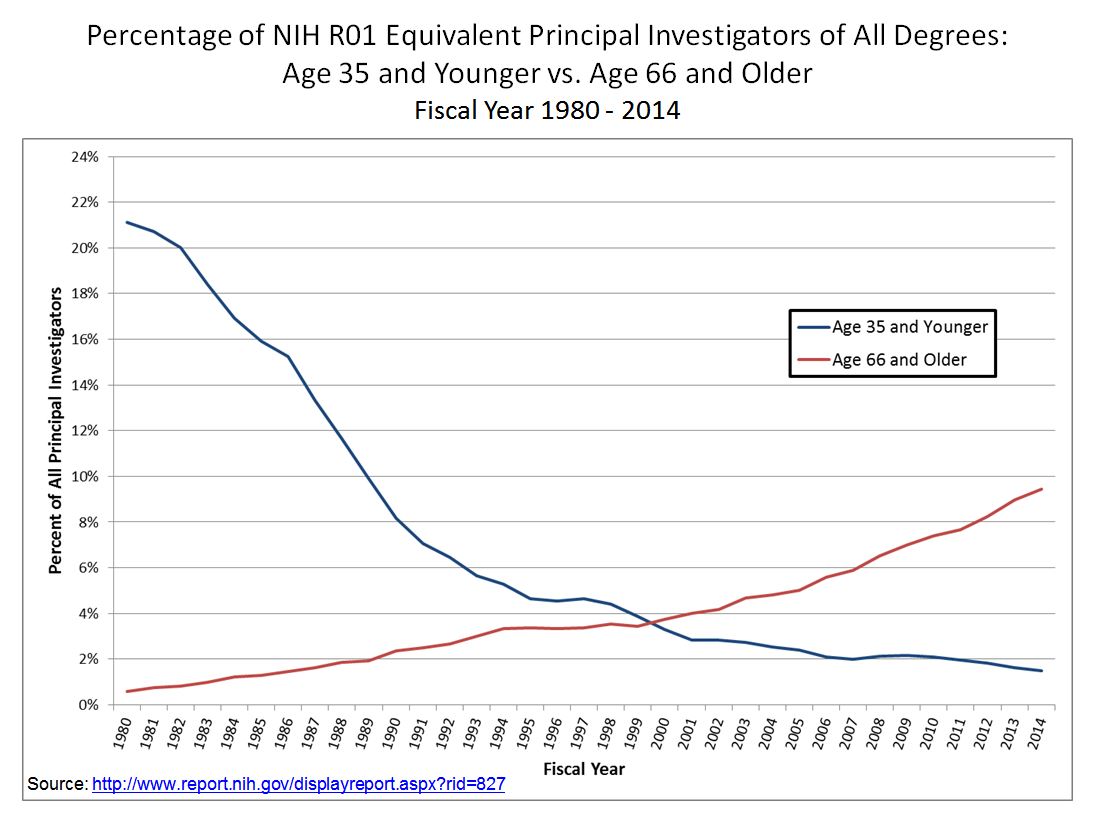
RPG = Research Project Grants. Source.
Another way to consider this gap is to simply plot the total share of R01 grants awarded to those under 35, compared to those aged over 65. The shift since 1980 is striking.
This age gap is perhaps most acutely felt among young researchers trying to access NIH funding for the first time. In 2020, the average age of a first-time R01 grant recipient was 44, up from 40 in 1995, and 34.3 in 1970.55 It is quite rare these days for researchers to get funded in their early 30s, and nearly unheard of to get funded in their 20s.
A 2017 paper56 found that NIH grantees are aging across the board. This demographic trend is most strongly felt in basic research: Since 1980, application rates for basic science-oriented grants have steadily fallen for researchers under 46 (almost a 40% drop between 1992 and 2014 alone), and steadily rose for researchers over 55.
Older Researchers Get Grants More Easily Than Younger Researchers
The NIH does not provide data on grant approval rates by age; only by broad categories. But we can extrapolate researcher age from these categories.
The NIH classifies “New” researchers as those who have yet to receive a standard NIH grant, like an R01. So new researchers can be anywhere from their late 20s to early 40s. In 2020, new researchers had a funding rate of 17.8%.
The “early stage investigators” (ESI) category is a subsection of new investigators who have completed a terminal degree within the past ten years. ESIs exclude graduate students and postdoctoral fellows, and are therefore on the older end of new investigators. In 2020, the funding rate for ESIs was 27.7%.
“Established” researchers have already received NIH grants. In 2020, the funding rate for “established” researchers was 32.9%.
Two researchers who I spoke with identified a phenomenon called the “valley of death.” According to them, NIH measures to boost grant acceptance for younger researchers have been reasonably effective, and well-established researchers still have their traditional advantages. But mid-career researchers are stuck in a “valley,” often struggling to get more grants to make the transition to the highest end of researcher establishment.
Why Are These Trends Happening?
Extramural grants are distributed by study sections, so NIH executives don’t have a great deal of direct control over who gets them. That makes the age issue all the more puzzling. Still, the NIH grant recipient demographic trends have been remarkably consistent.
There are six possible explanations.
First, the U.S. population is inherently getting older, on average. In 1970, 10% of Americans were 65 or older; in 1990, 12.5%; in 2010, 13%; in 2020, almost 17%.
Second, a major policy change is likely playing a role in the aging of American bioscience. In 1994, Congress amended the Age Discrimination in Employment Act to strike one of its few remaining age discrimination exemptions. 57 As a result, universities were prohibited from forcing faculty to retire at age 70. A 2021 study describes “dramatic effects” on the academic job market. 58 From 1971 to 1993, 1% of US faculty were over 70. From 1994 onward, 14% of faculty were over 70.
Third, as indicated by a few interviewees, postdocs tend to take longer to complete their work and advance to staff positions. Thus, there are fewer young researchers with the independence to apply for NIH grants.59
Fourth, older researchers have more existing grants than younger researchers, and it’s significantly easier to get grants renewed, rather than win first-time grants. 60
Fifth, there is a strong consensus that NIH grant applicants have become increasingly burdened by complex, arcane, bureaucratic rules (more on this in Part 4). Thus, established researchers with dedicated grant writing teams will have a systematic advantage over new researchers who have to learn all of these rules on their own.
Sixth, according to some papers61 and interviewees, study sections are biased against young researchers, perhaps because primary reviewers know who the grant applicants are, where they are in their careers, and thus may perceive older researchers as more competent.
Is The Current Preference For Older Researchers a Bad Thing For Science?
Twenty-somethings, in our society, can found multi-billion dollar tech companies, yet vanishingly few researchers under 30 ever receive funding from the NIH. Albert Einstein made his greatest breakthroughs in his late 20s; researchers with similar potential, today, are often confined to multi-year postdoctoral fellowships, but could have their own labs.
Evidence suggests that, in many fields, people do their best work in their 20s and 30s. This seems to particularly be the case in domains like physics, mathematics and chess, where there is a heavy dependence on fluid intelligence, which often peaks in an individual’s 20s.
Other domains, including bioscience research, also require the absorption of a massive corpus of work before any meaningful contributions can be made. Hence, perhaps great bioscientists tend to be older because they need time to not only amass all this knowledge, but integrate it in a manner that reveals potential breakthroughs. Great biologists and chemists have, anecdotally, tended to be older: Darwin, Mendeleev, Heisenberg, Pasteur. There are obvious exceptions: Watson and Crick were 25 and 37 when they discovered the helix structure of DNA. Rosalind Franklin was in her early 30s when she took Photo 51, the x-ray image that ultimately helped unravel DNA’s structure.
Funding more young researchers would likely result in more high-risk/high-reward projects, albeit with a higher failure rate.
Many researchers have fought against the NIH demographic trends out of reasonable career concerns. An NIH grant is nearly mandatory for a successful bioscience research career, so if fewer young researchers are getting them, then fewer careers are being launched. If this bottleneck is being instigated by purposeful or accidental biases that favor the status quo of established scientists, then young researchers are being marginalized by an unfair system.
On efficiency grounds, there are also concerns that the lack of young researcher funding might be stifling the next generation of talent. As more grants drift to older researchers, there are fewer opportunities for young researchers to advance in their fields, especially as older researchers continue to have longer careers.
Beyond raw research efficiency, one interviewee framed the issue as a matter of “passing the baton.” Researchers who continue their work into their 70s and beyond consume grants and resources which could start the careers of younger researchers. According to this interviewee, European governments rarely give grants to individuals (in any domain) over 65 for this reason.
Attempted, But Failed, Solutions
The NIH has taken measures to combat shifting demographics in grant recipients. Many have failed.
The NIH’s biggest campaign to address the age issue is their Early Stage Investigator (ESI) policy. Starting in 2009, and then overhauled in 2017 as the Next Generation Researchers Initiative, the program gave early researchers (defined as being within a decade of completing a terminal degree or medical school and having not received an NIH grant) an explicit advantage in grant evaluations.
“Applications from ESIs will be given special consideration during peer review and at the time of funding,” according to an NIH announcement. “Peer reviewers will be instructed to focus more on the proposed approach than on the track record, and to expect less preliminary data than would be provided by an established investigator."62
More concretely, ESIs get a bump on the NIH “payline.” When NIH extramural grants are submitted, they are scored and arranged by percentile; a specific percentile, such as 20%, is chosen as the payline. All applications below that payline are awarded. ESI policy varies by institute, but they tend to get a payline bump of 4 to 5%. So if the payline is 20%, then an ESI applicant may only have to be ranked in the top 25% of applications to get approval. 63
So how effective was the ESI program? Consider this graph:
The trend lines indicate that the share of grant money going to young researchers stopped declining shortly before ESI began for some reason. After ESI was implemented, the share increased slightly and then resumed its decline, albeit at a slower pace than before. It was clear that ESI was not creating a significant reversal in the trend as hoped, so many researchers continued to pressure the NIH for additional reforms.
In 2017, the NIH overhauled ESI with the Next Generation Researchers Initiative. Since then, the number of ESI researchers funded increased from 978 in 2016 to 1,412 in 2020, while their grant funding rate (for R01s and equivalents, the standard NIH grants) rose from 23.6% to 27.7%. So there seemed to be some progress, but…
Over the same time frame, the funding rate for researchers categorized by the NIH as “established” rose from 28.6% to 32.9%. And the funding rate for “new” investigators (at least one year of faculty experience), rose from 16.1% to 17.8%.64
So the ESI rate rose by 4.1%, the established rate rose by 4.3%, and the new investigator rate rose by 1.7%. Meanwhile, the NIH’s budget rose from $31.3 billion to $40.3 billion, or 29%.
Other initiatives, such as the Early Reviewer Career (ERC) program, have been more successful. One of the reasons researchers choose to serve on study sections is that it gives them first-hand insight into how to play the application game. But study sections are composed, largely, from researchers who have already received NIH grants. ERC invites new researchers (with at least one year of faculty employment, some publications, and no NIH grants), to sit in on study sections and learn how the review process works.65
Still, it’s surprising that ESI did not balance the scales for younger, NIH applicants.
Failed Reforms and Brute Force Solutions
Given the NIH leadership’s indirect control over the grant distribution process, many factors could have played into the continued concentration of grants among top researchers over the past five years. But there’s not an easy answer.
The ESI and the Next Generation Researchers Initiative were likely insufficient restructurings of the NIH’s extramural process, according to sources interviewed for this report. Francis Collins and the NIH’s leadership could have pushed harder, but they may have been discouraged from doing so by older researchers.
A typical researcher has to attend college for undergraduate studies, then a PhD or medical school, followed by a postdoctoral fellowship – often, a grueling 10 or more years of schooling. After all that, most researchers would be lucky to become a junior faculty member at a university or a low-level researcher at a private company, where they’ll have to work under established researchers for probably another six or more years before getting a decent shot at an independent research lab.
For individuals in the middle of that system, it can seem unfair, even guild-like. I’m sure there are many brilliant postdoctoral researchers, or even PhD students, with exciting research ideas who have almost no chance of getting direct NIH funding, let alone control of their own labs. But for individuals who have already gone through the system and come out the other side, its many tiers act as a barrier to entry. Their prestige and salary are in large part built on going through that gauntlet. If a brilliant, 29 year-old PhD student could snag an NIH grant, he would be jumping the queue and undermining the career value of decades of education and training.
Thus, established scientists have a strong incentive to maintain a high concentration of grants for later-career researchers, and to use the standard track researcher system as a credential base for filtering out grant applicants.
As I discuss in Parts 2 and 7, the NIH’s leadership is under pressure from its beneficiaries to maintain the status quo. Therefore, I believe it’s plausible that concerted pushback by older researchers has hindered reform.
Reversing the Demographic Trend
So, if the ESI and Next Generation Researchers Initiative isn’t helping young researchers enough, what can the NIH do to reverse the demographic trend?
The most direct method is to establish a new grant program with age or credential caps. If the NIH wanted to get really aggressive, it could produce more grants specifically for graduate students and postdoctoral fellows.
One interviewee pointed out that dumping more money into postdoctoral researchers is great policy from an efficiency perspective, because postdocs are cheap. Their salaries typically top-out around $70K, which is almost a third of what PIs make on NIH grants. The NIH could directly fund some of the most ambitious postdoctoral fellows at a low cost, rather than rely on normal grants trickling down to them. Fully-funded postdoctoral fellows could then contribute their skills to other labs, or even break away and start some of their own work.
While this would undoubtedly bring money to younger researchers, I’m unsure of the secondary effects. For instance, would major universities throw finite research space and funds behind younger researchers? How would older researchers feel about their brightest assistants abandoning them for their own work, before climbing the requisite academic ladder? It’s hard to say, but more ambitious efforts to fund young researchers could be transformative for biomedical research as a whole.
Part 4: Extramural Research
How Study Sections Work
While researching this report, study sections – the peer review bodies that decide which extramural grants receive funding – swiftly emerged as peoples’ most hated topic related to the NIH.
The core complaint is that study sections have an incentive problem; there simply isn’t a reason for study sections to fund the best research.
Compare the NIH study section to comparable research evaluation groups at pharmaceutical or biotech companies. In the latter case, there are concrete goals: pharmaceutical companies fund research that leads to quantifiable financial or clinical outcomes. Companies that pick the right research topics will thrive; others will fail. Individual grant evaluators in these institutions are rewarded or punished accordingly through financial payouts.
NIH study sections have no output-based success metrics, nor are there rewards or punishments based on success or failure rates. Study sections are not disbanded if they make poor decisions.
To be clear: those that serve on study sections are not lazy or apathetic. Numerous interviewees emphasized a sense of scientific integrity, a genuine desire to fund the best research. But NIH study section reviewers are, first and foremost, fallible human scientists. They are prone to prioritizing those grant submissions that support their own research.
Some interviewees consider this lack of a reward system to be a feature, not a bug. The absence of clear metrics, they say, permits a more open consideration of research proposals.
But consider the problem in economic terms. Like the NIH, study sections have no competition, no means of evaluating success and no consequences for success or failure. Therefore, they lack reasons to tighten up waste and optimize efficiency. Scientific integrity can mitigate emerging structural problems, but the negative feedback conveyed by interviewees, as well as the current academic literature, indicate that it is not enough to prevent the system from drifting further into mediocrity.
Study Section Structure and Process
Study sections are run by Scientific Review Officers (SROs), established scientists hired by the NIH. The SROs appoint a team of 10 to 30 volunteer researchers, to serve as reviewers on their study sections. Each study section is focused on narrow domains within a given NIH institute.
About six weeks before a study section meeting, each reviewer is assigned (on average) 7 to 10 grant applications on which they are the primary or secondary reviewers. Each application has two or three primary reviewers. The reviewer assigns a score (1 to 9, 1 being the highest) to each of these applications, based on a rubric designed for the study section.
All reviewers can read the other 50-100 grant applications, though they aren’t expected to.66
The SRO and the appointed reviewers of study sections are public knowledge. However, the primary reviewers on each application are not public knowledge, so applicants are not supposed to know who their primary reviewers are (whether that’s the case in reality is debatable). The names, academic histories, and current institutions of each grant applicant are known to the reviewers. The NIH extramural grant evaluation process stresses an emphasis on the project proposals, not the research institution or past accomplishments. The degree to which this guideline is followed is also debatable.
Two to three days before the study section meeting, the SRO posts all the primary and secondary reviewer scores and written evaluations on a confidential website. All reviewers in the study section have access to this website. Applications in the bottom half of the score distribution are not normally discussed at the upcoming meeting, though individual reviewers have the prerogative to “rescue” these proposals and bring them up during the meeting.
Study section meetings usually last two days, but can be done in one. The applications are evaluated one-at-a-time, and begin with the three primary reviewers giving five minute presentations. Then, the entire study section discusses the application for five minutes to an hour. With input from the primary and other reviewers, a score range is set by the SRO, and all reviewers submit a score.
The SRO writes up a summary of the evaluation for each grant, which includes the final averaged score. This score is converted to a percentile, and all applications below a certain percentile are funded. This percentile is known as the “pay-line,” and typically ranges from 15-30%.
Grant evaluations are sent to the applicants, but are not publicly available.
“Groupthink” and Other Issues
One of the recurring themes in interviewee descriptions, often explicitly stated, is that study sections tend to fall into “groupthink.” Theoretically, peer review panels are supposed to put independent voices together to argue different views until a consensus emerges, but often these groups of presumably highly intelligent and honest scientists succumb to group dynamics which prioritize conformity and conflict avoidance at the expense of objectivity.
The structure of the review process shoulders a lot of the blame. Recall: Roughly 10-30 scientists are given dozens, or even more than one hundred, grant applications to review. Each grant is assigned to two or three primary reviewers who lead the evaluation process.
Realistically, there are simply too many grant applications for everyone to read them all. Or in the words of one interviewee, “nobody wants to read all that shit.” Another interviewee admitted that they forced themselves to read all the grant proposals at their first study section, but then realized it was a waste of time and never did so again. Thus, most reviewers just read the applications that they will lead the discussions on, and then maybe skim a few others, or more commonly, read a few abstracts.
When it comes to voting on the other applications, most reviewers will naturally defer to the opinions of the two or three primary reviewers, thereby negating much of the value of gathering dozens of qualified scientists together for the peer review process in the first place. This establishes a consensus around each grant application which most reviewers rarely disrupt, whether out of a cost-benefit judgment that there’s nothing to gain by doing so, or out of apathy/laziness. One interviewee estimated that 90% of scores on a given application will be within 10% of the three principal interviewers' scores.
Numerous interviewees expressed frustration at this process, and felt like it was completely pointless to speak up for or against any grant application on which they weren’t the principal reviewers. One interviewee said that there is an unwritten rule that a reviewer can speak up for one, and only one, application that they’re not a primary reviewer on per session. If they speak up for more than one, they cross a social boundary and become that person who’s somewhere between an annoying do-gooder and boorish grandstander.
With so much evaluation power in the hands of two or three randomly chosen reviewers, lots of interviewees felt the study section process was highly arbitrary. If a grant happens to have a “charismatic” or forceful primary reviewer that likes the grant, its odds of approval go up considerably. One interviewee said they had seen mediocre grants get approval because an “alpha male” had basically bullied enough other reviewers. Other interviewees said they had seen good applications get shot down because their primary proponents were awkward, bad public speakers, or were non-native English speakers.
The conformity naturally induced by study sections is greatly amplified by what numerous interviewees called “politics.” Study sections are composed of experts in a single domain at various points in their careers. While this layering is intended to create multiple perspectives, it often leads to junior researchers deferring to senior researchers, either because they respect more experienced researchers, or because they are afraid of jeopardizing their careers by starting conflicts. More than one interviewee said they had seen blatantly mediocre grant applications get funding because everyone in the room was afraid to disagree with a prominent scientist. One interviewee claims that they were told in confidence by another reviewer that their application was torpedoed by a colleague who held a grudge. There is technically an appeals process for such issues, but it is considered cumbersome and unreliable.
An anonymous comment on an NIH article reflected the sentiments of the most negative interviewees: 67
“It is well known that NIH ‘confidentiality’ [of the primary reviewer to the grant applicant] is anything but, and a young PI risks career and reputation if they shoot down big names (not all, but there is a mafia of sorts). I’ve sat on panels, I’ve seen the influence from afar. Young PIs fall over themselves to get it good with the power brokers. I’ve seen young PIs threatened when they mentioned quietly that Big Boss X has data that is wrong. Some fields are worse than others, but it is overall a LOT uglier than most would believe.”
On the most nefarious end, a few reviewers mentioned, or implied, manipulation. Recall that study section reviewers are appointed by a Scientific Review Officer (SRO) who is hired by the NIH. The SRO can appoint pretty much whomever they want within basic qualification standards, so a few interviewees suggested that SROs are likely to appoint allies or lackeys, or dole out appointments as tokens of favor. Thus, the SRO might have enormous indirect influence over what sorts of applications are approved, even favoring some factions of research over others. One interviewee called this “turfism,” or the protecting of one’s research branch, and many other interviewees noted similar phenomena.
The most well-known example of turfism is the “Alzheimer’s Cabal.” As reported by STAT in 2019, for thirty years the National Institute of Neurological Disorders and Stroke (NINDS) was essentially captured by a faction of Alzheimer’s researchers who prioritized amyloid-based treatments and encouraged NINDS to avoid funding any other research pathways. One NINDS grant applicant related that a program officer “told me that I should at least collaborate with the amyloid people or I wouldn’t get any more NINDS grants.” Amyloid-based treatments for Alzheimer’s have come under heavy scrutiny over the last few years and the research field as a whole has drifted to more promising avenues.68
Granted, none of my interviewees mentioned anything as blatant as the Alzheimer’s Cabal, but they alluded to similar corrupt practices. One interviewee claims to have found “scams” whereby groups of researchers in particular fields purposefully wrote more grants to signal to the NIH that more funding was needed in that domain. The interviewee said this could be the result of a coordinated effort between the study sections and major researchers in the field, or it could be a decentralized trend that naturally emerges from the incentives.
Hearing these stories, I wondered how it was possible for an SRO to be so explicitly biased. There are at least two relevant failsafes in place: anonymous reviewing and SRO independence.
First, the study section process is theoretically anonymous since grant applicants can’t identify their primary reviewers, though the SRO and reviewers are public knowledge. But one interviewee said anonymous reviews are a “myth.” Though it violates NIH guidelines, reviewers can and do talk outside the confines of the review process, and primary reviewers can be identified if a grant applicant is so inclined to speak to the right people. After all, both the grant applicants and the reviewers are all in the same field, and likely know each other from conferences, papers, and general industry gossip. Petty grudges between individuals or factions can penetrate the allegedly anonymous review process, manifesting as blocked or artificially boosted grant applications.
Second, SROs are theoretically independent, and once appointed, are free to direct their review process however they see fit within limits. But the independence of SROs is at least questionable. One interviewee maintained that they genuinely are independent, much to the annoyance of NIH officers who wish they had more influence over loose cannon SROs. But another interviewee claimed that there was a lot more informal influence over SROs than is typically acknowledged. This could come in the form of the grant evaluation guidelines handed down to the SROs, or even from personal discussions behind closed doors between SROs and colleagues or NIH personnel. But again, no names were named, and it’s difficult to determine if such practices are widespread or occasional indiscretions.
One “No” is Enough to Kill a Grant
Grants that get funded are ultimately chosen by two or three scientists appointed by an SRO. The fickle, arbitrary nature of this process is readily apparent when compared to fundraising in the private sector.
Assume you have a startup company, and are looking for venture capital funding. There are hundreds of firms to choose from, and it’s a safe bet that you’ll be rejected by dozens of them (as many of the most successful startups are). A few firms will be true “believers,” though, and give you funding. The “reviewers” in this example are typically independent experts who make their living spotting and funding promising companies, rather than business people who run their own — potentially competing — companies in the field.
If you started a new supermarket chain, with the ambition of capturing a large part of the market, it’d be ludicrous if investments in your company were reviewed by Amazon, Walmart, and Target executives. Alas, this is precisely how NIH study sections work, whereby reviewers directly working in the field dictate grants for others and determine whether others will be able to enter and do work in that field.
Why do Researchers Serve on Study Sections?
Serving on a study section is not compulsory. Many of my interviewees complained about the time and effort required, and considered the process itself to be somewhere between badly flawed and farcical. So why do researchers choose to accept their appointments to study sections?
First, there is an implicit, professional obligation. Nearly all study section reviewers are current or former NIH grant recipients, and recipients are expected to serve. To my knowledge, there are no explicit mechanisms for punishing researchers if they receive a grant and choose not to serve, but I assume it would be frowned upon.
Second, it’s a great networking opportunity. Researchers meet and mingle both during the review process and outside working hours.
Third, it’s a good way to keep up with the current state of research in one’s domain. The submitted grants often indicate a field’s trends.
Fourth, nearly all interviewees noted that serving on a study section is the single best way to improve their own grant application odds. One interviewee said it was “extremely unclear” what reviewers were looking for until they served on a study section. Reviewers get to see the process from the inside, note guidelines, observe group dynamics, and therefore sharpen their own grant applications.
Fifth, it looks good on a resume.
Sixth, a few interviewees said they genuinely enjoy the process. Yes, there are a lot of headaches involved, but there can also be great discussions on the most important scientific issues of the day with brilliant people.
Application Response Times
The final study section complaint is that the NIH takes too damn long to respond to grant applications. The standard wait-time for a grant response, as both noted by interviewees and online sources, is three months, followed by another few months before the money is distributed. Most institutions take half as long to process a grant. Interviewees attributed the long turn-around time to a multitude of bureaucratic measures, which one source summarized as “some government-y reasons.”
For many labs, the long turn-around time creates a treadmill effect whereby researchers have to carefully juggle multiple, concurrent grants and grant applications without accidentally leaving funding gaps. As one interviewee described it:
“A lab applies for an NIH grant. It waits 5-6 months from submission to get the final result, which is sometimes ambiguous and requires a few more months for acceptance or rejection. If rejected, the lab will want to resubmit the grant since with the feedback from the study section it’s easier to get an acceptance the second time. But the lab must wait “eight or nine” months before they are allowed to resubmit. Then once they resubmit, they need to wait another 5-6 months for a response.”
In sum, it can take 14 months before a grant submission results in funding. Meanwhile, existing grants will be running out. Postdocs will be coming and going from the lab. New discoveries will pull research in one direction while other research paths peter out. PIs will have to take all of this complex management and budgeting into account while going through a process which typically takes 6-14 months to pay out.
Proposed Reforms to Study Sections
Proposed improvements to NIH study sections vary widely. Each proposal could be elaborated upon, but I’ll stick to short summaries:
Professional Model: Some interviewees suggested transforming study sections into full-time review boards staffed by NIH employees. This model should insulate the peer review process from “political” concerns in the academic sphere, like interpersonal or factional conflicts. Two interviewees spoke favorably of their experience with pharmaceutical companies using this method, and another pointed to the Federal Drug Administration as a good model. Theoretically, the NIH could even devise some sort of incentive structure based on the Success Metrics for a professional study section to aim for, though all must be wary of Goodhart’s law.
Randomness: A few interviewees suggested injecting randomness into the grant evaluation process, through a variety of means. For one such proposal, consider the previous breakdown: 100 applications, 50 are mediocre, 30 are good, 20 are excellent, and 25 must be chosen. A study section could use its normal methods to filter out the 50 worst applications, then choose to award 25 of the remaining 50 applications at random. Or they could choose 12 of the remaining 50 by regular methods, and then choose 13 at random.
With decent Success Metrics in place, over time the NIH could even compare the outcomes of selected and random grantees and see who does better. Randomness could obviously lead to worse results, but it could also eliminate a lot of the pettiness and politics of the selection process.
Intramural Model: One interviewee suggested that researchers should go through the normal study section system for their first grant, but then go through the far more streamlined intramural system for grant renewals. This means that, rather than design proposals for grant renewals to be reviewed by study sections (a time-consuming process for all involved), the researcher could get a site visit and update NIH grant managers on their progress.
People Over Projects: A popular alternative among interviewees is to reorient the grant selection process around “people, not projects.” Currently, the study sections take grant proposals based on individual projects, but other research institutions, like the Howard Hughes Medical Institute (HHMI), select applications primarily based on professional background and future potential. Supposedly, this system eliminates a lot of the minutiae which tends to amplify the bureaucratic processes of the NIH grant system.
Non-Academic Reviewers: One interviewee suggested that NIH study sections should be open to researchers from biotech and pharmaceutical companies. He blamed many of the study section errors on academic insularity, including a general risk-aversion. He claimed study sections are “biased against the very kind of research which is critical to the future of the enterprise.” Involving people from the private sector would also better connect the NIH to the marketplace and possibly push for more application-based research, though there is disagreement on whether this is a positive. At the very least, bringing in industry would likely add more dynamism to the overtly-academic setting. Another interviewee suggested mixing academic domains in the study sections to break factional strangleholds and make research proposals more broadly legible.
Give Grants to Institutions: Two interviewees suggested that, rather than give grants to researchers who are then supported by institutions, the NIH could give some or all grants to institutions or departments directly and then let them dole out the money. One interviewee was particularly concerned about the hierarchy within universities, though, wherein senior researchers controlled the grant money and had near-dictatorial powers over junior researchers. He suggested that by giving the institutions and the departments funding instead, there would be more oversight for this relationship and a fairer distribution of both funds and responsibility.
Randomized Controlled Trials (RCTs): The NIH could implement a variety of evaluative structures, let them run for an extended period of time (maybe 5-10 years), and then compare the results with Success Metrics.
In Defense of Study Sections
While all of my interviewees had critiques about study sections, a few were net-positive and made some good arguments.
One defended study sections by referencing the classic Winston Churchill quote: “It’s the worst form of peer review except for all the others that have been tried.” They argued that most of the flaws of study sections derive from the fact that they are run by humans who need to make judgment calls, and therefore there will always be such errors no matter how much the structures are tweaked.
Another interviewee speculated that many of the harsher critics of study sections were probably researchers who had their proposals repeatedly shot down, and so they’re a bit like a struck-out baseball batter complaining about the umpire.
A third interviewee acknowledged the cronyism and politics of study sections, but said there was a strong cultural resentment of these practices which discouraged them. In their words, there is a “constant struggle, but one where science is prevailing.”
This sentiment was reflected by quite a few interviewees (both the net-negatives and positives). They claimed that the greatest strength of study sections was a genuine commitment to science and that, while mistakes are made and politics is a reality, most researchers are passionate about their work and want to fund the best projects possible. One interviewee said the “degree of [virtue] is incredible and exciting.”
Transparency of Funding Decisions
Perhaps the only universally-praised aspect of NIH study sections is their transparency. When study sections evaluate a grant application, whether approval is granted or not, their score and notes are given to the applicant. This gives the applicant information on how to improve their application for the next attempt, both in specific aspects that apply to their current project, and in general aspects that apply to future projects. Some interviewees noted that sometimes they disagreed with the feedback (or even found the reasons for their rejection irrational), but generally they considered the feedback immensely helpful. It’s also worth noting that the grant application processes at other institutions (non-profits, etc.) rarely give feedback like the NIH.
Grant Writing and Maintenance
A common complaint among extramural grantees is that the NIH’s grant application process is onerous, bureaucratic, and time-consuming. On the most extreme end, some interviewees said that grant applications and maintenance make up a substantial portion of their general work time and reduce their overall research capacity.
Grant Preparation
The time and effort commitment for NIH grant writing varies by institution. Some universities and research labs provide considerable administrative support to researchers, including full-time administrators who handle part of the application process, particularly for well-established researchers. The help provided by top-tier universities, then, further supports the incumbents.
Other institutions provide little-to-no support. When asked how long it takes to write an NIH grant proposal, the responses I got ranged from weeks to one month of full time work at the lowest end, to three months at the highest end. One survey of bioscience researchers found that 57% of respondents spent one quarter of their work time writing grants.69
For well-established labs with a decently large staff, it’s common to have professional grant-writers who do nothing but write grants year-round. After all, a large lab will juggle multiple concurrent grants, and will constantly have to apply for renewals and new grants. So, a significant portion of money and manpower intended for scientific work will be permanently diverted to asking for more money. In one extreme case, an interviewee claims to know of a lab that spent 50% of its man hours to this end.
Many interviewees referred to grants being filled with paperwork that had no bearing on their experiment. In one case, an interviewee said they wrote a grant with 24 pages of scientific designs and 76 pages of “planning and maintenance” forms.
In both the low and high estimates, the interviewees referred to grant writing as a full-time job. As in, 40 hour weeks of nothing but planning and writing, with little time for ongoing research or teaching. An NIH criticism written by former NIH Director Harold Varmus and others described the problem:
”…biomedical scientists are spending far too much of their time writing and revising grant applications and far too little thinking about science and conducting experiments… Today, time for reflection is a disappearing luxury for the scientific community. In addition to writing and revising grant applications and papers, scientists now contend with expanding regulatory requirements and government reporting on issues such as animal welfare, radiation safety, and human subjects protection. Although these are important aspects of running a safe and ethically grounded laboratory, these administrative tasks are taking up an ever-increasing fraction of the day and present serious obstacles to concentration on the scientific mission itself."70
Grant Maintenance
Once an NIH grant is attained, there are requirements for periodic reports to the NIH, which are generally referred to as “grant maintenance.” The burden of this grant maintenance falls to varying degrees on researchers and institution administrators, depending on both.
Generally, universities have a larger administrative staff, which can carry a smaller or larger percentage of the burden. Additionally, established researchers will generally get more support from the administrations or, failing that, use their grant money to hire full-time grant maintenance workers.
This is one area where the NIH gets a gold star. Its grant maintenance is considered minimal, and usually consists of just one or two reports per year. Most other institutions have more requirements.
The worst example I heard of was, surprisingly, DARPA, an agency which usually gets glowing reviews. Two interviewees with DARPA grants said they had to write numerous overlapping period reports (i.e. quarterly, annually, at certain benchmarks, etc.). In one absurd instance, they had multiple period reports fall on the same due date, but they still had to write the same exact information on multiple forms and submit them simultaneously.
“Grantsmanship”
Some interviewees described a process called “grantsmanship,” or the contouring of grants to meet NIH evaluation standards. Grantsmanship can be helpful, annoying, or actively harmful to science as a whole.
On the most helpful end, as already mentioned, grant applicants adjust their applications based on feedback they get from study sections after failed applications. Most of the time, this advice is genuinely good and makes their projects better.
In the annoying middle, applicants adjust their applications based on feedback in ways they consider arbitrary. Maybe they need to stress a factor in their experiment they don’t find meaningful, or adjust their formatting in a way that their particular study section prefers due to pointless NIH regulations.
In the arguably harmful middle, some researchers heavily pursue fads in research. At any given time, some research topics are more likely to get funding than others because they are more popular in the scientific or mainstream communities. Many interviewees brought this up, though there was disagreement over whether it’s a bad thing. At worst, chasing fads causes valuable long-term research priorities to be abandoned for the sake of increasing grant application success odds.
On the definitely harmful end, some interviewees believe that grant applications across the board are leaning into a variety of negative trends as applicants sacrifice grant quality for higher acceptance rates. The most commonly cited trend is toward conservatism, where applicants will prune their project’s aims, focus on concrete deliverables, and avoid open-ended questions more characteristic of basic science. Some interviewees believe this sentiment has infected nearly all grant applicants, especially younger ones, but plenty of other interviewees were less pessimistic.
On the most extreme end, one interviewee said, “there’s not an honest submission” anymore, because virtually all applicants will lie to some degree about their research goals. Either they will write an application for a project they don’t really want to do but will do anyway for career purposes, or they will write an essentially fake application and then secretly follow their own research designs.
Part 5: Indirect Costs
The NIH’s extramural grant system is somewhat convoluted, but it has a logic. It is designed to promote private research with government money while simultaneously fostering research institutions through subsidies.
When the NIH awards extramural grants to researchers, the funds are divided between direct costs and indirect costs.
Direct costs are funds given to the researcher. These funds cover costs that are easily associated with the researcher and their team, including salaries, supplies, equipment, and lab space.
Indirect costs are funds that the NIH gives to the hosting institution (e.g.. a university, hospital, biotech company, etc.). These funds cover costs incurred by the hosting institution which are not easily identifiable with individual researchers or projects, such as depreciation and debt on research buildings, equipment depreciation, and “operation maintenance” (e.g. utilities, repairs, janitors, etc.).71 The indirect cost rate is a fixed percentage negotiated between the hosting institution and the government which determines how grants are divided between direct and indirect costs.
Consider a simple case: You are awarded a $1.5 million grant and your university’s indirect cost rate is 50%. Then, the NIH would send you $1 million to spend at your discretion, and another $500,000 directly to the university. (There are also some direct costs that don’t garner indirect costs, like equipment, so it’s usually a little more complicated to calculate.)
In 2017, the average standard institutional indirect cost rate was about 53%. In 2020, American universities earned about $8.1 billion in indirect costs. 72
Why are there indirect costs to begin with? Well, it’s because research — and especially bioscience research — is expensive. Universities house the researchers, give them lab space, give them equipment, manage them administratively, assist with grant applications, assist with networking, and shoulder a large share of total research costs. Without external funding, most federally funded researchers would be net-costs for the universities, and major research institutions couldn’t possibly employ as many researchers as they do today.
Most people that I spoke to for this report had criticisms of the indirect cost system, at least in its current form. One interviewee called the system “weird and inefficient.” Others called it “a complete rip-off” and “welfare for elite institutions.” At the most extreme end, others described it as a “scam” in which major universities with enormous endowments continually get taxpayer funding through an opaque and highly gameable system.
Less vocal interviewees said the NIH was probably overpaying many institutions through indirect costs and, at the very least, there is probably an incentive misalignment at play to the detriment of taxpayers.
Most people don’t know that top research universities earn a significant portion of their revenue from the government, ranging from about 10-30%. Most people also don’t know that their tax dollars are being used to fund expensive buildings and administrative staffs on campuses of universities with their own multi-billion dollar endowments. And most people don’t know that the flow of money from the government to these research universities has increased tremendously over the past thirty years, both due to the federal funding levels and the expansion of universities.
At the very least, there needs to be more scrutiny into the opaque, indirect cost system, and how universities attain and use government funds.
At its founding, the NIH and its predecessor institutions had no indirect costs. The NIH introduced the system in the 1950s on the grounds that large institutions with larger budgets were privileged since they were more easily able to sustain the administrative costs incurred by NIH grants. Initially, the indirect costs were capped at 8%, but soon rose to 20%. In 1965, the federal government began negotiating indirect costs with institutions. In 1991, the cap rose to 26% for administrative costs, but not for other indirect costs which remain uncapped.73
From 1967-1988, the share of NIH extramural spending on indirect costs increased from 17.1% to 32.6% (Note: A 50% indirect cost rate is equivalent to roughly 33.3% of the total grant going toward indirect costs).74 From 1998 to 2014, the share consistently hovered a little over 30%.75 By 2017, it had risen to one-third. In other words, about one-third of the NIH’s extramural budget, and almost 25% of its total budget, is being given directly to research institutions, primarily universities.76
In 2017, the average indirect cost rate was about 53%.77 However, individual rates vary considerably. Some examples (all on-campus, school year starting in 2020 or 2021 unless otherwise specified):
-
Massachusetts Institute of Technology (2022) – 55.1%78
-
University of California Los Angeles (2019) – 56%79
-
Stanford University – 57.7%80
-
California Institute of Technology – 68.4%81
-
Harvard University – 69%82
Note that private research foundations generally have even higher indirect cost rates. The Fred Hutchinson Cancer Research Center is at 76%,83 the Salk Institute for Biological Studies is at 90%,84 and the Cold Spring Harbor Laboratory is at 92%.85
Some of the increasing indirect cost rates are partially to blame on the NIH because of its increasingly burdensome grant requirements. The process of writing and maintaining grants requires an entire university bureaucracy, which means more salaries, more benefits and more employees.
How are Indirect Cost Rates Calculated?
A common misconception is that indirect cost rates are calculated by the NIH. Rather, most indirect costs paid to organizations for research projects are calculated by one of two “cognizant agencies;” the Health and Human Services Division of Cost Allocation handles most science matters (including the NIH), while the Department of Defense’s Office of Naval Research handles military projects.
These agencies negotiate indirect cost rates every four years with all institutions that house federally funded researchers. Once a rate is set, it applies to all grants given by a particular federal agency to the institution.
Basically, the government tries to figure out what percentage of total research costs for a given grant will be shouldered by the institution. Then, when it crafts grant payments, it divides the funds between a direct payment to the researcher and a payment to their institution.
The “pool,” which represents the institution’s costs, is determined by a negotiation between the cognizant agencies and the receiving institution. For the sake of relevance and simplicity, I’ll assume the institution is a university for the following sections.
The pool is all administrative and facilities costs incurred by the university in connection to its federally-funded researchers. Recall that administration costs are capped at 26%, and facilities costs are uncapped.
Again, the basic premise of reimbursing institutions for shouldering some of the costs of federally-funded researchers makes sense. But with the current system, there’s a potential issue:
As the NIH’s own instructional video points out,86 an old research building on a university campus generates low depreciation and probably has no debt, so it yields few subsidies. Since debt and depreciation are “pool” costs, such a building yields few indirect cost subsidies. A new research building generates high depreciation and debt, so it garners large subsidies. Thus, universities can often construct expensive, new research buildings at relatively small cost to them.
There have been a few instances of high-profile, indirect cost fraud. In 1994, Stanford University agreed to repay the US Navy $3 million for inflated indirect costs, including expenditures on flowers for the university president’s home and depreciation on a yacht.87 In 2016, Columbia University settled a case with the government for $9.5 million after it admitted that administrators purposefully mislabeled 423 research grants as “on-campus” rather than “off-campus” to cash in on the significantly higher on-campus indirect cost rate (61% to 26%).88 In 2020, the Scripps Research Institute paid $10 million to settle a claim that it had used its NIH-funded researchers for non-federal research tasks, including “writing new grant applications, teaching, and engaging in other administrative activities."89
One interviewee, part of a lab at an elite research university that receives NIH funding, shared an anecdotal story about indirect costs. This particular researcher had a conflict with another research team, because that research team kept coming into their lab to take dry ice, without permission. My interviewee’s team contacted the university’s maintenance company (which is 100% owned by the university) and requested that they install a key card reader on the lab door to keep the other team out. The building had hundreds of these card readers already; surely, this was a cheap, simple request.
But the maintenance company told the research team that purchasing and installing the key card reader would cost over $10,000.
The interviewee then searched for the key card reader online and found one for $15. Installing and integrating the key card reader into the university system would add costs, but nowhere close to $9,985.
It’s impossible to entirely understand the accounting at play, but the logic is clear. When a university owns the maintenance company, that company can charge the university inflated prices. The university will be happy to pay because it gets all that money back through ownership, while shuffling its maintenance costs into its indirect costs, thereby garnering more subsidies from the federal government.
Are Universities Profiting from the Indirect Cost System?
They are almost certainly not directly profiting from indirect cost reimbursements, but they are almost certainly profiting through secondary benefits of federally-funded research and indirect costs.
In 2012, universities self-reported that they spent $13.7 billion of their own funds on research. Out of that figure, $8.9 billion was initiated by the university, either as self-funded research or as funding in conjunction with federally-funded research. That left $4.6 billion as “unrecovered indirect costs,” or money the university paid to support research initiated by external grants which was not reimbursed by the granter.
Though the source doesn’t break the figure down further, it could be disaggregated between grants from the NIH, other federal sources, and non-federal sources. At top schools, the NIH typically accounts for 40-60% of federal grant money, and non-federal sources account for less than 10% of all grant money, so we can make a ballpark estimate that universities paid around $2 billion out of pocket for NIH research. In 2012, all university research spending was about $66 billion, 90 and the NIH’s extramural budget was about $22.3 billion. 91
It should be noted that the initial $13.7 billion figure is outdated (from 2012) and is based on self-reporting, so it has not been independently verified.
But assuming the figure is accurate, it indicates that universities are losing money from NIH research, and maybe indirect cost rates should be raised.
However, a naïve view is that the universities are simply channeling government funding into scientific research. There are significant secondary effects and externalities at play. Those funds are about more than just research; federal funds cause universities to gain prestige, attract better faculty, and bring in more donations. This is where universities truly profit from federal research money and indirect costs.
These indirect cost payments should be thought of as a subsidy. The universities are not contractors bidding to provide a service to the government (though the NIH does do a small amount of contracting). Rather, universities are generating their own research through their employees, and the NIH is providing the funding, which is only bounded by the skill of the university researchers in applying for grants and the capacity of the university to host them.
Because these subsidies scale to costs, universities are incentivized to keep increasing their costs, all the better to bring in more grants, boost their prestige, bring in better faculty and so forth, ad infinitum. Administrative costs are capped at 26%, so their scaling is controlled, but the facilities costs are uncapped.
We know that universities are responding to these incentives due to their revealed preferences. For instance, the rate at which universities created new biosciences doctorates perfectly conforms to the trends in NIH funding. In other words, as the NIH budget increased, universities built more research facilities and created more bioscience graduates, likely despite losing more money to indirect cost overflows. Clearly the secondary benefits were worth these costs.
Nearly everyone interviewed for this report supports increasing the NIH’s budget, but universities and their beneficiaries have a strong incentive to promote and expand the NIH.
On the other end of the equation, there is little incentive to restrict NIH expenditure, nor to keep careful track of its funds, since the federal agencies that set indirect cost rates are largely unknown to the general public, and suffer from similar incentive problems. The Department of Defense’s Office of Naval Research is unlikely to suffer repercussions if a university inflates its official facilities expenditures with fraudulent purchases.
I am not suggesting anything nefarious; but the current structure does align incentives of the NIH and large universities.
As an example, here are the budgets of five major NIH grant recipient universities, and the fraction of federal funds that comprise their total budget. In all of these cases, NIH grants account for approximately 40-60% of the university’s federal revenue.
The [University of California San Francisco] received $771 million in federal grants and contracts in 2019 out of $7.6 billion in total revenue; that’s 10% of the budget.92
[Harvard University] generated 11% of its revenue from federally-sponsored grants in 2020.93
[Yale University] received $618 million in federal grants and contracts in 2020 out of $4.3 billion in total revenue; that’s 14% of the budget.94
The [University of North Carolina Chapel Hill] received $722 million in federal grants and contracts in 2020 out of $2.2 billion in total revenue; that’s 33% of the budget. Additionally, the NIH was UNC’s single largest source of funding at $523 million, or 24% of total revenue.95
[Johns Hopkins University] received $2.5 billion in federal grants and contracts in 2019 out of $6.4 billion in total revenue; that’s 39% of the budget.96 97
Recall, however, that those research institutions with the highest indirect costs are private; the Fred Hutchinson Cancer Research Center has 76% indirect costs; it’s 90% and 92% for the Salk Institute for Biological Studies and Cold Spring Harbor Laboratory, respectively. These high, indirect cost rates support the notion that universities do subsidize research out of their own pockets, and might be doing so to a great extent.
Are Indirect Cost Rates Too Low?
In 2017, Peter Thiel met with NIH Direct Francis Collins to discuss possible reforms. Thiel identified rising indirect cost rates as an issue, and suggested caps. In an email exchange after the meeting, Collins said he was sympathetic to reform measures, but defended the foundations of the indirect cost system:
“Universities complain that NIH’s indirect cost rates don’t actually cover all the costs, and they have to make up for that with tuition, donations, state funds (if they have any), and endowment funds. In 2012, the Council on Government Relations (COGR) estimated that institutions put $13.7 billion of their own funds into subsidizing research.”
The $13.7 billion figure refers to institutional funds, or all the money universities spend on research. Those funds are composed from institutionally financed research, cost sharing, and unrecovered indirect costs.
Out of the $13.7 billion universities spent on research in 2012, $8.9 billion is university-initiated spending that does not subsidize federally-funded research, while about $4.6 billion is accounted for as the “unrecovered indirect costs.” This is the amount of money universities report as spending on subsidizing all federal (not just the NIH) research.
Impact of Indirect Costs on Researchers
Recall that the federal, indirect cost rate for top research universities varies between about 50% and 80%. This creates a strong incentive for universities to encourage their researchers to pursue specifically NIH grants.
Entire training seminars for grant applications, according to interviewees, were oriented around the NIH. Getting a few NIH grants is generally considered to be a requirement for promotion and tenure. Researchers can and do apply for other grants, but the default assumption is that university bioscience researchers should apply for NIH grants, as opposed to grants from non-profits which pay far lower indirect cost rates.s.
However, NIH grants may be preferred simply because they are the easiest ways for researchers to get lots of money. As mentioned, it’s nearly impossible for bioscience labs to run on non-profit grants outside of a select few sources, like HHMI.
On the other hand, there is definitely evidence that university administrators are motivated by indirect cost rates. In the most severe cases, some universities levy a “tax” on researchers for obtaining grants from non-NIH sources. In the case of Columbia University, the tax is equal to the NIH indirect cost rate minus the other institution’s indirect cost rate. That percentage is taken out of the researcher’s direct costs. This policy dramatically diminishes the value of non-NIH grants.
For example, if a Columbia University (62.5% NIH indirect cost rate) researcher receives a grant from the Bill and Melinda Gates Foundation (10% indirect rate), with a direct cost of $1 million and a total value of $1.1 million, a whopping $525,000 intended for the researcher is redirected to the university, bringing the direct cost benefits to the researcher down from $1 million to $475,000. And frankly, that’s underselling the transfer, because non-profit grants tend to be much smaller than NIH grants; once the tax is paid, the remaining money might not even be worth applying for.
Slush Funds and Aggregations
There’s a quirk in the indirect cost system that makes it either more fair, or more distortionary, for certain universities. There is, in effect, a secondary cost layer embedded within the indirect costs.
It works like this: Universities receive lump sums of cash from the indirect costs paid by the NIH upon the disbursement of a grant to a university researcher. The university administrators then divide the funds between two categories of costs: fixed and discretionary. A portion of the funds go toward building depreciation, electricity, maintenance, and any long-term, fixed costs. Another portion of the cash is spent at the discretion of administrators on laboratories, according to their needs.
That second category resembles direct costs, except the funds are controlled by administrators, rather than researchers. The administrators might buy equipment for an individual lab, shift administrative resources for grant writing, or even refund some of the indirect costs back to the researcher.
If this system works effectively, then it could be a very strong safeguard against distortionary effects of the indirect cost system. Consider:
If no money was spent on discretionary, indirect costs, labs that spend less money would effectively be subsidizing labs that spend more money. All bioscience labs at a given university, after all, pay the same NIH indirect cost rate, regardless of how much equipment, electricity and maintenance they use. For instance, depreciation and debt on research buildings are indirect costs, so smaller research teams indirectly pay for a disproportionately larger share of the building debt and depreciation.
I initially assumed that the indirect cost system would tend to benefit experienced researchers at the expense of inexperienced researchers, since the former typically has more funding, larger labs, and uses more equipment. But the opposite might be more common.
Universities often spend huge sums on labs, equipment, or “cores” designed for common use to attract new researchers, while older researchers are left with older labs. Also, one interviewee claimed their administrators tend to provide more support to lesser-funded researchers with indirect costs, while better-funded labs are expected to take care of themselves.
This second example reveals a key factor in this secondary cost layer. How well this layer works is highly dependent upon the skill and objectivity of university administrators. Good university administrators could shift indirect cost benefits in a manner commensurate with each researchers' resource consumption. Bad administrators could fail to line up the costs and benefits, or worse, shift the benefits in an arbitrary manner, perhaps to support particular researchers for non-scientific reasons, like university politics.
Unfortunately, I did not get the opportunity to study this aspect of the indirect cost system in-depth. The wise dispersion of indirect costs within universities could be widely sound, corrupt, or highly variable by university. For what it’s worth, the interviewee who knew the most about this spoke highly of the administrators he knew at multiple universities.
How Do Indirect Costs Affect University Hiring?
Because of indirect costs, universities view researchers as grant-generating machines. At least from a financial standpoint, their value to the university is measured in how much money they can pull in. Output, or research quality, can impact prestige or maybe fundraising, but it is relatively devalued compared to the funds brought in by awarded grants.
Universities pass this incentive along to their faculty. Researchers are under immense pressure to generate grants; failure to do so puts their job at risk.
This could have a strong, negative effect on the tenor of university research. I’m sure faculty want to do good work, but they probably want to keep their job more. So most university researchers will sacrifice research quality for higher odds of grant acceptance and guaranteed funding.
Potential Reforms
In his email with Peter Thiel, NIH Director Francis Collins discussed potential reforms for indirect costs and claimed that eliminating indirect costs entirely would have “devastating consequences.”
Specifically, “American bioscience would take a giant step back,” as most research universities would close down, the rest would drastically cut their operations, and research diversity would plummet as a handful of surviving institutions would absorb NIH funding.
Collins then considered a 20% flat indirect cost rate, but said it would have “major negative effects.” He suggested 40% would be better, but still quite bad, especially because it would penalize specialized, private research institutions that tend to have high indirect cost rates (80%+).
Collins’s favored reform proposal, at least in this email chain, would be to reduce all current indirect costs rates by 5% (so a 50% rate would go down to 47.5%). Collins stated that this would cause institutions to “tighten their administrative belts” without serious issue, and would free up quite a bit of NIH money.
Part 6: Intramural Research
The NIH’s intramural program is highly regarded. It was portrayed by many interviewees as one the best places in the United States to conduct bioscience research; they said it is “encouraging of innovation,” “very nourishing,” “an incredible concentration of both talent and ideas.” Another interviewee said that they received “unprecedented support” for an ambitious project that would have taken “10 to 20 years” to get started at a university.
Private industry has a profit motive that discourages basic science research, academia is rife with politics and career climbing, and private foundations often don’t have enough resources to pursue transformative work. But the NIH’s intramural program has a massive budget; researchers are given nearly complete freedom to pursue ambitious projects. And while extramural researchers probably get more raw funding, a dollar in an NIH lab goes a lot further than a dollar at a top research university, and thus NIH researchers are most likely better-funded in real terms.
Much of this research freedom is derived from the intramural program’s streamlined review process.
To become a researcher at the NIH, most people are selected through an application process. Reviews are done internally, by NIH personnel. Once hired, a researcher is given a budget, staff, and a lab in NIH facilities. They can work on almost any project for the next four to five years. After that time, a site review occurs, during which the researcher retroactively justifies their use of funds and outlines their research progress. One interviewee described this process as being narrative-oriented; intramural researchers often must connect and talk about multiple, disparate research projects.
If approval is granted after this site review, the researcher’s budget is replenished and they can continue working for another four- to five-year term. Theoretically, this process is indefinite. Some internal NIH grants also allow a PI to leave the intramural program to work elsewhere for a set period of time, with guaranteed extramural funding for a few years.
The NIH intramural programs often receive applications from the best postdoctoral researchers, according to several interviewees, because of this research freedom and minimal bureaucratic overhead.
NIH Salaries are Not Competitive
Given the above, why doesn’t everyone want to work at the NIH?
Because NIH intramural pay is mediocre to terrible compared to anywhere else.
A new intramural NIH researcher’s (PI's) salary is typically in the $140,000 to 180,000 range, depending on the specific institute. The ceiling for most researchers is around $220,000, though some can push a bit above $300,000 if they generate income through cuts from intellectual property royalties.98
The NIH is located in Bethesda, Maryland, a suburb of Washington D.C. In 2019, Bethesda’s median household income was $164,000 (2.5X the national median) and the median house value was $911,000.99 Its county, Montgomery County, is the 18th wealthiest in America. This actually makes it one of the poorer D.C. suburbs, since four of the ten wealthiest counties are nearby.100 101
After ten or more years of intensive schooling, a salary of $150,000 in one of the most expensive parts of America is a tough sell. Many excellent researchers consider it too big of a sacrifice, especially as they get older and, potentially, start a family.
NIH intramural researchers are often highly qualified, and so their academic options, too, are elite.
Another option for bioscience researchers is to enter the private sector. One source, who used to make about $180,000 as an NIH researcher, consistently found jobs at private medical practices that offered more than $500,000 per year.
This experience was comparable for two other people that I interviewed. One source, who largely lauded the NIH, recently left their intramural program because the pay was not enough to support a family.
If a researcher is willing to forgo significant money for the sake of working at the NIH, then they are likely to be genuinely committed to scientific progress. In other words, perhaps the low salaries are a filtering mechanism that eliminates all but the most passionate and scientifically-driven applicants.
A pessimistic perspective, expressed by several sources for this report, is that the NIH intramural program employs mostly mediocre scientists. Talented scientists will take their big paychecks in the private sector, they say, or opt for the prestige of universities over the NIH.
Federal Employee Regulations
NIH researchers are federal employees. With that title comes regulations and overhead:
-
Employees must give six months notice for any business travel (such as going to a medical conference, giving a presentation at a university, and so forth).
-
All business travel must be coordinated through a government agency if the researcher wants reimbursement. This is convenient for NIH employees since the agency buys the tickets and automatically reimburses them, but due to some obscure regulation, the agency can only handle trips between the office location and one destination. This means that NIH employees can’t do multi-leg trips. If an employee wants to go to a conference in New York and then another one immediately afterwards in Boston, she must fly from Baltimore to New York, then back to Baltimore, and then to Boston.102
-
There are caps on many expenses, such as hotels. As a result, major conferences will often set aside cheaper, often overbooked hotel rooms for federal employees.
-
Federal employees need permission to attend “WAGs,” or widely attended gatherings. This includes basically any meeting, conference, or dinner where a private entity, like a pharmaceutical company, gives a material good to attendees (such as coffee and donuts). The approval process takes months, unless it’s a major event that has pre-approval.
-
Federal employees can’t apply for many private grants because their rules don’t align with federal regulations. For instance, a federal employee can’t get a grant that transfers any IP rights to the granting agency, because that violates federal rules that dictate IP rights to researchers.
-
Federal employees can’t take government-provided equipment out of the country (such as laptops).
-
Federal employees cannot accept gifts of more than $20. Conference swag is prohibited, then, unless an employee wants to try to calculate the value of a Pfizer coffee mug.
One interviewee referred to these regulations as “death by a thousand cuts.” He said that he basically never goes to sponsored dinners, never accepts gifts and never attends conference exhibits.
That same interviewee was sympathetic to the regulations, though, and said they were put in place to avoid conflicts of interest and avoid undue influence on federal employees by private entities.
Detractors of Intramural NIH Research
While most interviewees were overwhelmingly positive about the NIH intramural programs, there are detractors.
Two sources said they knew of intramural researchers who did very little useful work because they were so bogged down in red tape and federal regulations, or said that the NIH failed to attract top-tier, highly motivated researchers because of low pay.
The NIH’s intramural system, said another source, had been the gold standard for scientific research in the 1950s and 60s, but has institutionally ossified. They claimed that the much-vaunted freedom of researchers was just as likely to produce laziness and coasting as brilliant scientific breakthroughs.
And, indeed, the NIH is uncharacteristically opaque about intramural research. Every extramural grant is extensively detailed (recipients, institutions, monetary amount, description, and so forth) on the NIH’s RePORT website, but there is no comparable database for intramural research. Thus, it is nearly impossible to evaluate the intramural program’s success, at least for outsiders.
Part 7: Political Influences
There is little correlation between NIH funding levels and general disease burden on American society, according to two studies from 1998 and 2011.103 As a federal agency, embedded within the Department of Health and Human Services, political influence is inevitable. This influence, according to sources interviewed for this report, is driven by major universities, top research beneficiaries, and nonprofits that lobby for specific diseases.
Too Much Trend Chasing
NIH research priorities are affected by public opinion and usually reflect an increased awareness of particular diseases. The National Cancer Institute has been the highest-funded NIH institute for some time, largely due to political forces prompting President Nixon’s “War on Cancer.”
More than one interviewee claimed that such funding swings have caused some inefficiency in the long-run, particularly with too much money going to cancer and heart disease, and thus resulting in more, lower-yield research being funded. As mentioned, many other interviewees brought up the tendency of some extramural researchers to pursue fads in their grant applications to increase grant approval odds.
Or Maybe Not Enough Trend Chasing
At other times, the NIH has perhaps been too resistant to public opinion.
Consider March 2020 and the onset of the COVID-19 pandemic in the U.S. While millions of people were infected with a novel virus, Patrick Collison, Silvana Konermann, and Tyler Cowen found that the speed with which the NIH shifted federal funds for COVID-19 research was too slow. Few new projects were being funded and many top scientists were waiting for permission from the NIH to repurpose existing funding for the worst pandemic in modern history.
In April, the trio started a program called Fast Grants, under the auspices of the Mercatus Center at George Mason University. Throughout 2020, Fast Grants raised $50 million and disbursed 260 grants. Each grant application took about 30 minutes to complete, and was approved or rejected within 48 hours by a panel of 20 experts who reviewed more than 6,000 applications. Funded projects include an accurate, saliva-based COVID-19 test, successful clinical trials for a generic drug which might reduce COVID-19 hospitalization rates by 40%, and significant research into long COVID. 32% of grant recipients said their research was accelerated by “a few months,” while 64% of recipients claim they would never have been funded without Fast Grants.104
During the first three months of the pandemic, the NIH spent a mere $2 million on COVID research. One intramural researcher complained, in an interview, that their entire project was defunded for new COVID research.
Still, the NIH has a serious problem with rapidly changing research priorities. In 2020, the agency spent $2.4 billion (5.7% of the total budget) on COVID research, including less than $800 million on clinical research. By January 2022, that figure climbed to $5 billion.105
These might seem like small values, but the NIH's budget is usually determined years in advance. Most of its grants extend over several years. A fixed set of funds are allocated for fellowships and grants. With all this money already being set aside then, it’s probably a fair assumption that only a fraction of the NIH’s discretionary budget can be doled away for a new public health crisis. Directing money in a near-sighted way, too, can ultimately impinge upon long-term research.
The sluggish response of this public health agency to a massive public health problem is a serious cause for concern. With a $40+ billion budget, the NIH should have some latitude for quickly redirecting funds even as, according to one interviewee, many researchers switched their projects to COVID-related aims without informing the NIH of their reprioritization until later.
Lobbying by Universities and Nonprofits
NIH beneficiaries can and do lobby the NIH, the Department of Health and Human Services, and Congress. These expenditures are public record.
In 2020, the NIH received 486 registered lobbyists representing 179 clients. Most clients are disease-related nonprofits, such as the American Lung Association and the Tourette Syndrome Association. Others are broader class advocacy groups, like the AARP and National Nurses United.
Many lobbying clients are universities, particularly those that receive lots of NIH funding. Of the top 50 largest NIH recipients in 2020 (including universities and private labs), 40 institutions106 engaged in lobbying of the NIH through the Association of American Universities or the Association of Public Land Grant Universities.107
Combined, the Association of American Universities108 and the Association of Public Land Grant Universities109 spent $1.4 million on lobbying in 2022, though the expenditure was spread out among a dozen federal agencies.
Research!America is a lobbying group devoted to championing the NIH (and to a lesser degree, other public health agencies).110 In 2020, it spent $3.7 million, almost a third of which went to its five main employees.111 The group is financed by many of the same organizations hiring the other lobbyists, including 133 universities and affiliated institutes112 and most of the top 50 beneficiaries.113 114
United for Medical Research is a similar group that lobbies the NIH on behalf of universities. Its funders include both the Association of American Universities and the Association of Public Land Grant Universities, as well as a handful of other major NIH recipients (Princeton, Vanderbilt, Washington University in St. Louis, Harvard, Johns Hopkins and others), some professional associations (ex. American Association for the Advancement of Science), some single disease advocacy groups (ex. the Alzheimer’s Association), and Johnson & Johnson.115 In 2020, United for Medical Research spent $120,000 on lobbying.116
Though I assume the influence of lobbying is relatively minor, given the small expenditures, lobbying directed at Congressmen may prove more effective. According to a 2014 study by professors from New York University and Columbia University, single-purpose nonprofits have a significant impact on which diseases Congress singles out for research recommendations to the NIH. The study found that lobbying led to a higher likelihood for Congress to put “soft earmarks” in bills, which encouraged the NIH to establish particular funding priorities. The NIH, according to this study, has increased funding on rare diseases between 3-15% as a result.117
Electoral Pressure
Congress appropriates the NIH’s funding, and universities benefit from this funding. Large research universities have tens of thousands of employees, massive endowments, and plenty of influential connections. They are often among the largest and wealthiest employers in Congressional districts. Thus, Congressmen from districts with large universities are likely more prone to support increased funding for the NIH.
Consider Johns Hopkins University**,** the largest recipient of NIH funds in 2020. In 2019, JHU employed 27,300 people and the Johns Hopkins Health System, a nonprofit that oversees multiple hospitals and medical centers, employed an additional 23,470 people. These are the two largest, private employers in the Greater Baltimore area and the 7th Congressional district. 118
The University of California Los Angeles was the fourth-largest recipient of NIH funds in 2020. That year, UCLA had 51,000 employees, making it the fourth largest employer in Los Angeles County, with almost 10,000 more employees than the largest private employer.119
Even if Johns Hopkins and UCLA are not directly giving money to Congressmen, many of their thousands of employees are voters, many donate to political campaigns, and others respond to overtures from academic leaders.
Nearly all of the top NIH beneficiaries have 10,000 or more employees, with the notable exceptions of a few private institutes, like the Fred Hutchinson Cancer Research Center (over 3,500120) and the Research Triangle Institute (almost 6,000121). But the importance of all of these institutions is undoubtedly amplified by their fame and influential leadership.
Other Instances of Influence on the NIH
Numerous interviewees, particularly those with former leadership experience at the NIH, stated that there was distortionary political pressure on NIH operations. They pointed to instances of powerful individuals picking up the phone and calling the right people.
One interviewee said he knew a Congressman who told him: “the NIH is [like the Mafia], and the only time in my life when I’ve been physically threatened is when I proposed reducing NIH funding." The same congressman also referred to the NIH as a “jobs program," at least from a congressional perspective.
Consider “The Art of Politics and Science,” a 2007 memoir by Dr. Harold Varmus, who served as director of the NIH from 1993-1999, and then as director of the National Cancer Institute from 2010-2015. Varmus describes being beset by special interest groups and politicians, who constantly angled for shifts in the budget to support their hobby horses.122
“One of my first exposures to this problem occurred soon after I arrived at the NIH, when I received a call from my own former congresswoman, Nancy Pelosi, asking me to add $50 million to the budget for AIDS research. As the representative from one of the districts most heavily affected by the epidemic, her wishes were understandable,” Varmus wrote. “I declined as politely as I could.”
Varmus continues:
“Sometimes it was not so easy to say no. Late one afternoon in May of 1996, as I was walking on the NIH campus, my driver pulled up with an urgent expression and asked me to take an emergency call on the car phone. A senior member of the administration—Jack Lew, then deputy director, later director, of the Office of Management and Budget—told me that the president had just met the recently paralyzed actor Christopher Reeve for the first time that afternoon and had promised in the presence of the press to increase spending on spinal cord research by $10 million. I started to explain the difficulties of doing this, when the phone was passed at the other end to a more junior person, who said, basically, just do it, don’t argue, or you won’t get the money. Of course, the White House was not in a position to send us any additional funds directly. But the president’s wishes are always obeyed. When the next accounting was made of disease-specific spending at the neurology institute (formally known as the National Institute for Neurological Diseases and Stroke, or NINDS), the funds for spinal cord research were accordingly higher, and funds for other purposes were proportionately lower.”
Single issue nonprofits, too, are a focus of Varmus' book. Specifically, he writes that advocates often “refuse to recognize, or to care, that funds for their disease must come from funds being spent elsewhere, including funds used for a disease important to another group of advocates.”
In one instance, nonprofit advocates for diabetes research “adopted an unusually militant approach,” Varmus writes, “openly criticizing my management of the NIH and even picketing the Illinois home of John Porter, the chairman of our House Appropriations Subcommittee. (The attack on Porter was ill-conceived: his wife was a diabetic, his concerns about the disease were evident, and his support for the NIH was unsurpassed.) … These groups can limit the possibilities for adjusting the distribution of funds across the institutes, since annual changes in each institute’s budget are scrutinized carefully, to one hundredths of a percentage point, and any downward deviation from the NIH average is likely to result in appeals to influential congressional or administration advocates. This has the sometimes stultifying effect of keeping institute budgets in approximate lockstep, simply to avoid the inevitable outcry if one of them doesn’t do well.”
Part 8: Concluding Thoughts
Size, Reproducibility and Intramural Funds
The NIH is, and will continue to be, the driving force behind American and global bioscience research.
No other organization comes close in scale or scope. The next largest bioscience funders are corporations focused on profit, which spend their money on clinical research for drugs developed based largely on NIH-funded research. Most foreign government agencies and nonprofits that fund basic research have an order of magnitude less funding. The only organizations that could compete with NIH funds are tech giants, but they are constrained by profit motives that prevent them from dumping shareholder dollars into unpredictable basic research.
Thus, the NIH is simply irreplaceable. If it had never existed, bioscience would be far behind its current progress. If it blinked out of existence, the industry would collapse. There are simply no organizations or forces that can fill its void. It is a shining example of proper government in action, an agency that puts money into public goods with tremendous positive externalities.
With great money comes plentiful flaws, however. The NIH produces plenty of good research, but that’s also a function of the sheer amount of its funding. Between 1990 and 2020, it spent more than a trillion dollars (in 2020 dollars).
And, in return for some good research, the NIH has become a gravity well that warps the entire bioscience research industry, seeding it with distortions and bad incentives. At best, the NIH has wasted an enormous amount of taxpayer money on low-value work; at worst, it is partially responsible for the monstrously inefficient and costly university system, it has inculcated an entire generation of scientists with bad habits and it has failed to upkeep standards in bioscience research.
The Reproducibility Project: Cancer Biology — a crowdsourced project — aimed to replicate cancer biology studies published between 2010 and 2012. But out of 23 high-impact studies, “fewer than half yielded similar results,” according to reporting by Science. The project originally flagged 53 papers for replication experiments, but “vague protocols and uncooperative authors” meant that just 23 could even be tested.
Perhaps these issues are limited to the NIH’s extramural program, though. The NIH itself — its intramural program — is possibly the best facility on earth for a bioscience researcher. Intramural researchers are given unprecedented access to resources and connections. They are free to pursue far-reaching ideas.
Working within the NIH, though, is rife with bureaucratic red tape. As a federal employee, many scientists carry out excellent work, but often at the cost of millions of dollars in personal income, at least in the long run, due to low government salaries. Most researchers would prefer to roll the dice with a biotech company, even if it means giving up the dream of basic research.
Researchers who remain in the NIH system are also dominated by entrenched interests – the major universities, top researchers and their labs – and often seek to join them. Ever since the Boom Decade from 1993-2003, when the NIH’s budget more than doubled, this government agency has been a covert feeding trough for some of the most lavish rent-seekers in the country.
Slow and Steady
Yes, the NIH has flaws. The study sections need to be reformed, grants should probably be more widely distributed, indirect costs are perhaps too high and more money should be put aside for basic science and high-risk projects.
But these are natural flaws endemic to any giant bureaucracy, especially in the government. Some are caused by rent-seekers, some by bureaucratic inertia, and some by necessary, but annoying, government safeguards designed to protect taxpayers and prevent conflicts of interest.
All things considered, the NIH leadership has done a remarkable job at containing these problems. They don’t have the murky autocratic power of a university president, nor the narrow profit goals of a private company leader, but suffice instead as appointees and hires of presidential administrations. NIH leaders quietly serve as stewards of scientific research, while appeasing whatever random noise comes from their political overlords. Often, that means making non-ideal compromises. And despite all that, the NIH’s budget has quadrupled in the last thirty years and the organization remains largely immune from political influence. Being a leader at the NIH is a thankless task, and it’s impossible to come away unscathed, but we should believe Francis Collins and his team have done as well as they have.
By juggling political influences and serving as ‘quiet’ leaders, though, impactful research often goes unfunded. There is, perhaps, a need for more trend-chasing at the NIH.
The NIH has sacrificed bioscience research dynamism in favor of a tedious, but safe slog. The United States and the world have probably missed out on countless medical breakthroughs because the most daring researchers and projects are never funded. A great mass of NIH-funded researchers, instead, produce work of incremental value. Researchers have accepted a Faustian bargain, whereby they play along with the system and lie on their grants for the sake of longevity in their career.
Ideally, ambitious researchers could go outside the system and get private funding, but the NIH has made that almost impossible. The entire industry has been warped to NIH standards. University labs and private foundations can’t survive without NIH money, so they too hire conservative researchers who are good at getting NIH grants, and those researchers train younger researchers to be conservative, and so on. A few lucky researchers can get money from the rare, well-run private institute, like the Howard Hughes Medical Institute, but there is precious little good money to go around.
Entrenched Interests
Famous researchers and universities derive lopsided benefits from the NIH, and that poses tough questions about the fair use of taxpayer funds. But these concerns are overstated by critics (many of whom feel personally slighted about not being a member of those same entrenched interests). The NIH has a responsibility to promote good science, and not to give away money in an arbitrary manner. Bioscience follows the 80/20 rule, just like everything else, so of course a fairly small percentage of researchers and universities will produce the best work and therefore will get the most NIH funds. To do anything else would be putting politics ahead of science.
The NIH’s favoritism for entrenched interests has also solidified the most elite education system in history. The US’s combined research universities are incomprehensibly better than any other nation’s. Outside of maybe the U.K. and China, there are scant countries with even three universities comparable to the US’s top 20 schools. The NIH and other federal research agencies deserve a lot of credit for building these institutions to their current heights.
Perhaps the selective dealing of funds to the most influential universities, though, is not a good thing. Elite universities already have their own multi-billion dollar endowments. They are given further, taxpayer-backed dollars by the NIH to build laboratories and expand their bureaucracies for their own profit and prestige. These universities have gamed this system perfectly; armies of accountants, lawyers, and administrators have blown up school budgets and then expertly negotiated with the federal government to get more of it covered by the taxpayer through rising, indirect costs. When the Boom Decade ended, it was not the universities that suffered; it was the legions of researchers suckered into becoming cheap labor in their bloated research programs, despite grim job prospects.
These well-funded universities also coax political connections in Washington D.C. to ensure that the NIH maintains its status quo. Any whiff of real reform, whether to give more grants to younger researchers, cap grant amounts, or reduce indirect costs, is hindered by phone calls and popular backlash from rank-and-file researchers.
Meanwhile, the hapless NIH leadership is beset by do-gooder advocacy groups, lobbyists, and politicians who push the NIH to support one cause or another with no understanding of the big picture. The sway of political winds or lunch with the right Congressman could shift millions of dollars to one place or another over the objections of wise stewardship. And somehow, despite all this, when there is good reason to rapidly move funds around the NIH — such as when the biggest pandemic in modern history kills millions of citizens — red tape and lethargy prevent America’s largest public health agency from doing much to combat the problem for months.
In response to these issues, NIH supporters suggest incremental reforms which either never happen (like grant size caps), or do happen and then get immediately canceled (like the Grant Support Index), or they happen and don’t work (like the Next Generation Researchers Initiative).
Forward Facing
These issues are not enormous, nor are they impossible to solve. With sufficient political will, good leadership, and careful politicking, incremental reforms could curtail these issues.
We know these flaws can be fixed because the NIH is one of the vanishingly few government agencies that has legitimate esprit de corps. I personally haven’t talked to anyone who works at the Department of Energy or Transportation or Commerce, but I doubt they love their bureaucracies as much as many of my interviewees love the NIH. Many top scientists probably think that the NIH is the single best part of the U.S. government.
Best of all, we know an easy fix to many of the NIH’s problems: increase its budget.
More money means more funds for young researchers and smaller institutions. It means less arbitrary study sections. It means more specialized, high-risk grants. And best of all, it means more money going to invaluable bioscience research. A higher budget, though, should also be treated with care.
More money would alleviate some of the NIH’s problems, yes, but in the long run, it could make them worse. So many of the NIH’s modern issues were caused by its great influx of funds during the Boom Decade, which not only threw open its structure to rampant exploitation, but entrenched the NIH’s dominant role as a gravity well in distorting the rest of the bioscience research industry.
Giving the NIH more money could lead to more of the same. More low-margin research will be funded by a hopelessly broken study section system. The universities will take more money for their giant, new laboratories and colossal administrative staff. The entire bioscience industry will have an even stronger incentive to base all of its standards on the NIH. Bioscience as a whole could slow in the long run.
What bioscience needs is not more money placed into a giant centralized government bureaucracy that’s been dominating an entire field of science for three quarters of a century, but rather genuine reform. Maybe the NIH could be fixed, but that would require politically impossible maneuvers, like breaking it into smaller pieces, or allocating many of its grants randomly, or eliminating the entire study section system.
One alternative is a complete restructuring of the bioscience industry and the ushering in of new organizations and players to fill the NIH void — private companies, philanthropists, and more agile government agencies. Short term chaos is probably unavoidable, but in the long run, bioscience will be healthier without the NIH in its current form.
And despite all this — despite the duality of the NIH, its triumphs and failures — it is undoubtedly one of the great success stories of the American government. It is a federal institution that has invigorated a valuable public service and has built a vast wealth of knowledge that has translated into incredible inventions and the saving of countless lives.
Correction: An earlier version of this report mentioned, in the last paragraph, that the Scientific Management Review Board has not issued a report since 2010. Their last report was issued in 2015.
Do you have thoughts or feedback on this New Science report? Please send your thoughts to matt.faherty530@gmail.com and to alexey@newscience.org.
This report was written by Matt Faherty, an independent researcher and freelance writer. It was funded by the Emergent Ventures. It was edited by Niko McCarty and Alexey Guzey. Niko is a data journalist and former bioengineer, based in New York. Alexey is the Executive Director of New Science.
New Science is a 501c3 research nonprofit with the mission to facilitate scientific breakthroughs by empowering the next generation of scientists and building the 21st century institutions of basic science. If you’re interested in our work, feel free to see our one-year fellowship for young scientists, the rest of our site, and to subscribe to our updates.
We thank Samir Unni and Egan Peltan for helpful feedback on earlier drafts.
This report is licensed under Creative Commons Attribution 4.0 International License.
Cite this essay:
Faherty, M. “New Science’s Report on the NIH.” newscience.org. 2022 April. https://doi.org/10.56416/209ubx
Or download a BibTeX file here.
References
-
“H.R.6164 - 109th Congress (2005-2006): National Institutes of Health Reform Act of 2006.” Library of Congress. Accessed April 22, 2022. https://www.congress.gov/bill/109th-congress/house-bill/6164. ↩︎
-
“Database of ERC Funded Projects.” European Research Council. Accessed November 17, 2021. https://erc.europa.eu/projects-figures/project-database. ↩︎
-
“Annual Report 2020.” Bill and Melinda Gates Foundation. Accessed November 16, 2021. https://www.gatesfoundation.org/about/financials/annual-reports/annual-report-2020. ↩︎
-
“What is the IRP?” National Institutes of Health. Accessed November 20, 2021. https://irp.nih.gov/about-us/what-is-the-irp. ↩︎
-
“Budget.” National Institutes of Health. June 29, 2020. Accessed November 20, 2021. https://www.nih.gov/about-nih/what-we-do/budget. ↩︎
-
With the exception of pure clinical research which might be done solely through pharmaceutical companies. ↩︎
-
“Appropriations (Section 2).” NIH. Accessed January 15, 2022. https://www.nih.gov/about-nih/what-we-do/nih-almanac/appropriations-section-2. ↩︎
-
“U.S. Military Spending/Defense Budget 1960-2022.” Macrotrends. Accessed February 7, 2022. https://www.macrotrends.net/countries/USA/united-states/military-spending-defense-budget. ↩︎
-
Gugliotta, Guy. “USDA: Giant in Need Of Reform? Sixth-Largest Agency Was Las Restructured During the 1930s.” Washington Post. January 14, 1993. Accessed February 7, 2022. https://www.washingtonpost.com/archive/politics/1993/01/14/usda-giant-in-need-of-reform-sixth-largest-agency-was-last-restructured-during-the-1930s/952e0cf7-575b-43b5-9e3c-d18c0b334774/. ↩︎
-
“United States Department of Agriculture.” US Department of Agriculture. Accessed February 7, 2022. https://www.fsa.usda.gov/Internet/FSA_File/08budoutlays.pdf. ↩︎
-
“Interior Secretary Lujan Focuses On Continued Accomplishments In The Fiscal Year 1993 Budget.” US Department of the Interior Indian Affairs. January 29, 1992. Accessed February 7, 2022. https://www.indianaffairs.gov/as-ia/opa/online-press-release/interior-secretary-lujan-focuses-continued-accomplishments-fiscal#:~:text=Secretary%20of%20the%20Interior%20Manuel,generating%20employment%20and%20economic%20opportunities.. ↩︎
-
“Secretary Norton Lauds Congressional Support for Interior’s 2004 Budget.” US Department of the Interior. November 10, 2003. Accessed February 7, 2022. https://www.doi.gov/sites/doi.gov/files/archive/news/archive/03_News_Releases/031110a.htm. ↩︎
-
“Federal Budget Receipts and Outlays.” The American Presidency Project. Accessed February 7, 2022. https://www.presidency.ucsb.edu/statistics/data/federal-budget-receipts-and-outlays. ↩︎
-
Harris, Laurie A. “The National Science Foundation: FY2018 Appropriations and Funding History.” Congressional Research Service. December 18, 2017. Accessed February 7, 2022. https://sgp.fas.org/crs/misc/R45009.pdf. ↩︎
-
“NIH Awards by Location and Organization - NIH Research Portfolio Online Reporting Tools (RePORT).” NIH. Accessed April 22, 2022. https://report.nih.gov/award/index.cfm?ot=DH,27,47,4,52,64,41,MS,20,16,6,13,10,49,53,86,OTHDH&fy=2020&state=&ic=&fm=&orgid=&distr=&rfa=&om=n&pid=. ↩︎
-
Beardsley, Tim. “Where Science and Religion Meet.” Scientific American. February 1998. Accessed January 12, 2022. https://www.scientificamerican.com/article/where-science-and-religion-meet/. ↩︎
-
West, John G. “NIH Director Francis Collins Isn’t A National Treasure, He’s A National Disgrace.” Discovery Institute. October 8, 2021. Accessed January 13, 2022. https://www.discovery.org/a/nih-director-francis-collins-isnt-a-national-treasure-hes-a-national-disgrace/. ↩︎
-
Ollstein, Alice Miranda. “Anti-Abortion groups demand ouster of NIH chief over fetal tissue issue.” POLITICO. December 18, 2018. Accessed January 13, 2022. https://www.politico.com/story/2018/12/18/anti-abortion-groups-francis-collins-nih-1068724. ↩︎
-
Collison, Patrick; Hsu, Patrick; Cowen, Tyler. “What We Learned Doing Fast Grants.” Future. June 15, 2021. Accessed February 7, 2022. https://future.a16z.com/what-we-learned-doing-fast-grants/. ↩︎
-
Collison, Patrick; Hsu, Patrick; Cowen, Tyler. “What We Learned Doing Fast Grants.” Future. June 15, 2021. Accessed February 7, 2022. https://future.a16z.com/what-we-learned-doing-fast-grants/. ↩︎
-
“Discovering Life in Yellowstone Where Nobody Thought it Could Exist.” U.S. National Park Service. Accessed April 22, 2022. https://www.nps.gov/articles/thermophile-yell.htm. ↩︎
-
“NIH Director’s Pioneer Award.” NIH. Accessed January 20, 2022. https://commonfund.nih.gov/pioneer. ↩︎
-
“Evaluation of the NIH Director’s Pioneer Award Program‐DP1.” James M. Anderson. 2013. Accessed April 22, 2022. https://dpcpsi.nih.gov/sites/default/files/CoC-051413-Pioneer-Award-Program-DP1.pdf. ↩︎
-
“NIH Director’s New Innovator Award.” NIH. Accessed January 20, 2022. https://commonfund.nih.gov/newinnovator. ↩︎
-
“NIH Director’s Early Independence Award.” NIH. Accessed January 20, 2022. https://commonfund.nih.gov/earlyindependence. ↩︎
-
“NIH Director’s Transformative Research Award.” NIH. Accessed January 20, 2022. https://commonfund.nih.gov/tra. ↩︎
-
“NIH Thiel Communications.” Buzzfeed. Accessed February 8, 2022. https://www.documentcloud.org/documents/7203720-NIH-Thiel-Communications.html. ↩︎
-
Collins, Francis. “Testimony on the Transformative Power of Biomedical Research.” NIH. May 17, 2017. Accessed February 7, 2022. https://www.nih.gov/about-nih/who-we-are/nih-director/testimony-transformative-power-biomedical-research. ↩︎
-
“NIH’s Funding of Basic Research.” NIH. Accessed February 7, 2022. https://www.nih.gov/news-events/basic-research-digital-media-kit#:~:text=NIH's%20Funding%20of%20Basic%20Research,-A%20large%20part&text=Funding%20for%20basic%20science%20research,51%25%20of%20the%20NIH%20budget.. ↩︎
-
Lauer, Mike. “NIH’s Commitment to Basic Science.” NIH. March 25, 2016. Accessed February 7, 2022. https://nexus.od.nih.gov/all/2016/03/25/nihs-commitment-to-basic-science/. ↩︎
-
“Waste Report.” Rand Paul. Accessed January 3, 2022. https://www.paul.senate.gov/wastereport. ↩︎
-
“Sen. Rand Paul Uncovers $500,000.00 NIH Grant Studying People’s Fondness for Spicy Foods.” Rand Paul. Accessed January 3, 2022. https://www.paul.senate.gov/news/sen-rand-paul-uncovers-50000000-nih-grant-studying-people%E2%80%99s-fondness-spicy-foods. ↩︎
-
“Dr. Rand Paul Uncovers $380K NIH Study on ‘Freshman 15’ in Latest ‘The Waste Report’.” Rand Paul. Accessed January 3, 2022. https://www.paul.senate.gov/news/sen-rand-paul-uncovers-50000000-nih-grant-studying-people%E2%80%99s-fondness-spicy-foods. ↩︎
-
Bump, Philip. “The various dishonesties in Rand Paul’s cocaine-quail presentation.” Washington Post. May 28, 2021. Accessed January 3, 2022. https://www.washingtonpost.com/politics/2021/05/28/various-dishonesties-rand-pauls-cocaine-quail-presentation/. ↩︎
-
Wahls, Wayne P. “The NIH must reduce disparities in funding to maximize its return on investments from taxpayers.” Elifesciences.org. 2018. Accessed January 10, 2022. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5882298/pdf/elife-34965.pdf. ↩︎
-
Collins, Francis. “New NIH Approach to Grant Funding Aimed at Optimizing Stewardship of Taxpayer Dollars.” NIH. May 2, 2017. Accessed January 10, 2022. https://www.nih.gov/about-nih/who-we-are/nih-director/statements/new-nih-approach-grant-funding-aimed-optimizing-stewardship-taxpayer-dollars. ↩︎
-
Wahls, Wayne P. “The NIH must reduce disparities in funding to maximize its return on investments from taxpayers.” Elifesciences.org. 2018. Accessed January 10, 2022. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5882298/pdf/elife-34965.pdf. ↩︎
-
Wahls, Wayne P. “The NIH must reduce disparities in funding to maximize its return on investments from taxpayers.” Elifesciences.org. 2018. Accessed January 10, 2022. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5882298/pdf/elife-34965.pdf. ↩︎
-
“NIH Awards by Location and Organization.” NIH Research Portfolio Online Reporting Tools (RePORT). Accessed April 22, 2022. https://report.nih.gov/award/index.cfm?ot=&fy=2020&state=&ic=&fm=&orgid=&distr=&rfa=&om=n&pid=. Note: not counting “contracts, construction, and other.” ↩︎
-
“Top 50 NIH-Funded Institutions of 2020.” Genetic Engineering and Biotechnology News. September 21, 2020. Accessed January 10, 2022. https://www.genengnews.com/a-lists/top-50-nih-funded-institutions-of-2020/. ↩︎
-
I am excluding #6 Leidos Biomedical Research Inc. because it is a federally funded research and development center, practically an intramural contractor. ↩︎
-
Wahls, Wayne P. “The NIH must reduce disparities in funding to maximize its return on investments from taxpayers.” Elifesciences.org. 2018. Accessed January 10, 2022. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5882298/pdf/elife-34965.pdf. ↩︎
-
“Active Funding by State.” RePORTER. Accessed January 10, 2022. https://reporter.nih.gov/. ↩︎
-
Pier EL, Brauer M, Filut A, Kaatz A, Raclaw J, Nathan MJ, Ford CE, Carnes M. Low agreement among reviewers evaluating the same NIH grant applications. Proceedings of the National Academy of Sciences. 2018 Mar 20;115(12):2952-7. ↩︎
-
Nakamura RK, Mann LS, Lindner MD, Braithwaite J, Chen MC, Vancea A, Byrnes N, Durrant V, Reed B. An experimental test of the effects of redacting grant applicant identifiers on peer review outcomes. Elife. 2021 Oct 19;10:e71368. ↩︎
-
“List of countries by income equality.” Wikipedia. Accessed April 22, 2022. https://en.wikipedia.org/wiki/List_of_countries_by_income_equality. ↩︎
-
Nielsen MW, Andersen JP. Global citation inequality is on the rise. Proceedings of the National Academy of Sciences. 2021 Feb 16;118(7). ↩︎
-
Collins, Francis. “New NIH Approach to Grant Funding Aimed at Optimizing Stewardship of Taxpayer Dollars.” NIH. May 2, 2017. Accessed January 29, 2022. https://www.nih.gov/about-nih/who-we-are/nih-director/statements/new-nih-approach-grant-funding-aimed-optimizing-stewardship-taxpayer-dollars. ↩︎
-
Wahls, Wayne P. “The NIH must reduce disparities in funding to maximize its return on investments from taxpayers.” Elifesciences.org. 2018. Accessed January 10, 2022. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5882298/pdf/elife-34965.pdf. ↩︎
-
Wahls, Wayne P. “The NIH must reduce disparities in funding to maximize its return on investments from taxpayers.” Elifesciences.org. 2018. Accessed January 10, 2022. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5882298/pdf/elife-34965.pdf. ↩︎
-
Collins, Francis. “New NIH Approach to Grant Funding Aimed at Optimizing Stewardship of Taxpayer Dollars.” NIH. May 2, 2017. Accessed January 10, 2022. https://www.nih.gov/about-nih/who-we-are/nih-director/statements/new-nih-approach-grant-funding-aimed-optimizing-stewardship-taxpayer-dollars. ↩︎
-
Kaiser, Jocelyn. “Updated: NIH abandons controversial plan to cap grants to big labs, creates new fund for younger scientists.” Science. June 8, 2017. Accessed January 20, 2022. https://www.science.org/content/article/updated-nih-abandons-controversial-plan-cap-grants-big-labs-creates-new-fund-younger. ↩︎
-
Peifer M. The argument for diversifying the NIH grant portfolio. Molecular biology of the cell. 2017 Nov 1;28(22):2935-40. ↩︎
-
“Scientists worry about plan to cap individual labs’ federal funding”. Robert Weisman. Boston Globe. Accessed April 22, 2022. https://www.bostonglobe.com/business/2017/06/07/local-scientists-worry-about-nih-proposed-cap-funding-for-individual-labs/8qv6ydg4xBT32Y4a7LkVfN/story.html. ↩︎
-
Levitt, Michael; Levitt, Jonathan M. “Future of fundamental discovery in US biomedical research.” PNAS. March 10, 2017. Accessed January 9, 2022. https://www.pnas.org/content/pnas/114/25/6498.full.pdf. ↩︎
-
Levitt, Michael; Levitt, Jonathan M. “Future of fundamental discovery in US biomedical research.” PNAS. March 10, 2017. Accessed January 9, 2022. https://www.pnas.org/content/pnas/114/25/6498.full.pdf. ↩︎
-
“Amendment of the Age Discrimination In Employment Act of 1967.” Illinois Periodicals Online. Accessed April 22, 2022. https://www.lib.niu.edu/1986/im861220.html. ↩︎
-
Ho DE, Mbonu O, McDonough A. Mandatory Retirement and Age, Race, and Gender Diversity of University Faculties. American Law and Economics Review. 2021;23(1):100-36. ↩︎
-
Levitt, Michael; Levitt, Jonathan M. “Future of fundamental discovery in US biomedical research.” PNAS. March 10, 2017. Accessed January 9, 2022. https://www.pnas.org/content/pnas/114/25/6498.full.pdf. ↩︎
-
“Are Attempts at Renewal Successful?” NIH Extramural Nexus. Accessed April 22, 2022. https://nexus.od.nih.gov/all/2016/02/16/are-attempts-at-renewal-successful/. ↩︎
-
Levitt, Michael; Levitt, Jonathan M. “Future of fundamental discovery in US biomedical research.” PNAS. March 10, 2017. Accessed January 9, 2022. https://www.pnas.org/content/pnas/114/25/6498.full.pdf. ↩︎
-
“NIH Funding Advantages To New and Early Stage Investigators.” Meg Bouvier. Accessed January 9, 2022. https://megbouvier.com/nih-funding-advantages-to-new-and-early-stage-investigators/. ↩︎
-
“NIH Paylines & Resources – Grant Q&A & advice.” Medical Writing, Editing & Grantsmanship. Accessed April 22, 2022. https://writedit.wordpress.com/nih-paylines-resources/. ↩︎
-
Lauer, Mike. “Data on Implementing NIH’s Next Generation Researchers Initiative.” NIH Office of Extramural Research. July 12, 2021. Accessed January 9, 2022. https://nexus.od.nih.gov/all/2021/07/12/data-on-implementing-nihs-next-generation-researchers-initiative/. ↩︎
-
“Early Career Reviewer (ERC) Program.” NIH. Accessed January 9, 2022. https://public.csr.nih.gov/ForReviewers/BecomeAReviewer/ECR. ↩︎
-
“How to Be a Member of an NIH Study Section.” HHMI. 2009. Accessed January 20, 2022. https://www.hhmi.org/sites/default/files/Educational%20Materials/Lab%20Management/study_section.pdf. ↩︎
-
“Age Distribution of NIH Principal Investigators and Medical School Faculty.” NIH Extramural Nexus. Accessed April 22, 2022. https://nexus.od.nih.gov/all/2012/02/13/age-distribution-of-nih-principal-investigators-and-medical-school-faculty/. ↩︎
-
Begley, Sharon. “The maddening saga of how an Alzheimer’s ‘cabal’ Thwarted progress toward a cure for decades.” STAT News. June 25, 2019. Accessed January 1, 2022. https://www.statnews.com/2019/06/25/alzheimers-cabal-thwarted-progress-toward-cure/. ↩︎
-
Collison, Patrick; Hsu, Patrick; Cowen, Tyler. “What We Learned Doing Fast Grants.” Future. June 15, 2021. Accessed February 7, 2022. https://future.a16z.com/what-we-learned-doing-fast-grants/. ↩︎
-
Alberts, Bruce; Kirschner, Marc W.; Tilghman, Shirley; Varmus, Harold. “Rescuing US biomedical research from its systemic flaws.” PNAS. April 22, 2014. Accessed January 2, 2022. https://www.pnas.org/content/111/16/5773. ↩︎
-
Rockey, Sally. “All About Indirect Costs.” NIH. September 11, 2015. Accessed January 4, 2022. https://nexus.od.nih.gov/all/2015/09/11/all-about-indirect-costs/. ↩︎
-
“NIH Awards by Location and Organization.” NIH Research Portfolio Online Reporting Tools (RePORT). Accessed April 22, 2022. https://report.nih.gov/award/index.cfm?ot=&fy=1992&state=&ic=&fm=&orgid=&distr=&rfa=&om=n&pid=. ↩︎
-
Rockey, Sally. “All About Indirect Costs.” NIH. September 11, 2015. Accessed January 4, 2022. https://nexus.od.nih.gov/all/2015/09/11/all-about-indirect-costs/. ↩︎
-
McLoughlin, William J.; Parker, Michael G. “Indirect Cost Rates what they are and how they Reached their Current Levels.” Cancer Investigation. June 11, 2009. Accessed February 15, 2022. https://www.tandfonline.com/doi/abs/10.3109/07357909009012079. ↩︎
-
Rockey, Sally. “All About Indirect Costs.” NIH. September 11, 2015. Accessed January 4, 2022. https://nexus.od.nih.gov/all/2015/09/11/all-about-indirect-costs/. ↩︎
-
Miller, Brian; Richardville, Kyle. “The NIH needs to become leaner and more innovative. Here’s how to do that.” STAT. January 22, 2019. Accessed January 4, 2022. https://www.statnews.com/2019/01/22/making-nih-leaner-more-innovative/. ↩︎
-
“NIH Thiel Communications.” Buzzfeed. Accessed February 8, 2022. https://www.documentcloud.org/documents/7203720-NIH-Thiel-Communications.html. ↩︎
-
“Current and Historical Facilities and Administration (F&A) Rates.” MIT. Accessed January 4, 2022. https://ras.mit.edu/facilities-and-administrative-fa-rates. ↩︎
-
“F&A Agreement.” UCLA. Accessed January 4, 2022. https://ocga.research.ucla.edu/facilities-and-administrative/. ↩︎
-
“Facilities and Administrative (F&A) Cost Rates.” Stanford. Accessed January 4, 2022. https://doresearch.stanford.edu/topics/rates. ↩︎
-
“Proposal Information Caltech.” Caltech. Accessed January 4, 2022. https://researchadministration.caltech.edu/osr/proposalinfo. ↩︎
-
“FAS Research Administration Services.” Harvard University. Accessed January 4, 2022. https://research.fas.harvard.edu/indirect-costs-0. ↩︎
-
“Facilities and Administrative Rates.” Extranet. Accessed February 8, 2022. https://extranet.fredhutch.org/en/u/osr/fa-rates.html. ↩︎
-
“Nonprofit rate agreement.” Federal Demonstration Partnership. Accessed February 8, 2022. https://fdpclearinghouse.org/organizations/305. ↩︎
-
“Nonprofit rate agreement.” Cold Spring Harbor Laboratory. Accessed February 8, 2022. ↩︎
-
Rockey, Sally. “All About Indirect Costs.” NIH. September 11, 2015. Accessed January 4, 2022. https://nexus.od.nih.gov/all/2015/09/11/all-about-indirect-costs/. ↩︎
-
Celis, William 3^rd^. “Navy Settles a Fraud Case on Stanford Research Costs.” New York Times. October 19, 1994. Accessed January 5, 2022. https://www.nytimes.com/1994/10/19/us/navy-settles-a-fraud-case-on-stanford-research-costs.html. ↩︎
-
“Manhattan U.S. Attorney Announces $9.5 Million Settlement With Columbia University For Improperly Seeking Excessive Cost Recoveries In Connection With Federal Research Grants.” The United States Attorney’s Office Southern District of New York. July 14, 2016. Accessed January 5, 2022. https://www.justice.gov/usao-sdny/pr/manhattan-us-attorney-announces-95-million-settlement-columbia-university-improperly. ↩︎
-
“The Scripps Research Institute To Pay $10 Million To Settle False Claims Act Allegations Related to Mischarging NIH-Sponsored Research Grants.” U.S. Department of Justice. September 11, 2020. Accessed February 8, 2022. https://www.justice.gov/opa/pr/scripps-research-institute-pay-10-million-settle-false-claims-act-allegations-related. ↩︎
-
“Higher Education Research and Development: Fiscal Year 2019.” National Science Foundation. Accessed November 16, 2021. https://ncses.nsf.gov/pubs/nsf21314#general-notes. ↩︎
-
“NIH Awards by Location and Organization.” NIH Research Portfolio Online Reporting Tools (RePORT). Accessed April 22, 2022. https://report.nih.gov/award/index.cfm?ot=&fy=2012&state=&ic=&fm=RP_SB,RP,SB,RC,OR,TR,TI&orgid=&distr=&rfa=&om=n&pid=. ↩︎
-
“2019 Annual Report.” UCSF Comptroller’s Office. Accessed February 8, 2022. https://controller.ucsf.edu/financial-statements/campus/2019/2019-annual-financial-report. ↩︎
-
“Financial Report Fiscal Year 2020.” Harvard University. Accessed February 8, 2022. https://finance.harvard.edu/files/fad/files/fy20_harvard_financial_report.pdf. ↩︎
-
“Budget Book Fiscal Year 2021.” Yale University. Accessed February 8, 2022. https://your.yale.edu/sites/default/files/fiscal-2021-public-budget-book.pdf. ↩︎
-
“Comprehensive Annual Financial Report.” UNC Finance and Operations. Accessed February 8, 2022. https://finance.unc.edu/wp-content/uploads/sites/298/2020/11/2020-comprehensive-annual-financial-report.pdf. ↩︎
-
“The Johns Hopkins University Consolidated Financial Statements.” Johns Hopkins University University Finance. Accessed February 8, 2022. https://finance.jhu.edu/reports_guides/financial_reports/AnnualReport2020.pdf. ↩︎
-
“Higher Education Research and Development: Fiscal Year 2019.” National Science Foundation. Accessed February 8, 2022. https://ncses.nsf.gov/pubs/nsf21314#general-notes. ↩︎
-
These figures were summarized by an interviewee. Full pay data can be found here: https://www.federalpay.org/employees ↩︎
-
“Quickfacts Bethesda CPD Maryland.” United States Census Bureau. Accessed February 6, 2022. https://www.census.gov/quickfacts/bethesdacdpmaryland. ↩︎
-
Burrows, Dan. “The 10 “Real” Richest Counties in the U.S.” Kiplinger. August 2, 2021. Accessed February 7, 2022. https://www.kiplinger.com/real-estate/603232/the-real-richest-counties-in-the-us. ↩︎
-
“Median Household Income In the Past 12 Months (In 2019 Inflation-Adjusted Dollars).” United States Census. Accessed February 7, 2022. https://data.census.gov/cedsci/table?q=B19013%20%20MEDIAN%20HOUSEHOLD%20INCOME%20IN%20THE%20PAST%2012%20MONTHS%20%28IN%202016%20INFLATION-ADJUSTED%20DOLLARS%29&g=0100000US%240500000&tid=ACSDT1Y2019.B19013. ↩︎
-
Note – The interviewee who told me about this wasn’t sure if it only applied to his institute or all institutes. ↩︎
-
Balaguru, Logesvar; Walsh, Christi; Dun, Chen; Kung, Christopher; Meyer, Andrea; Migliarese; Makary, Martin. “NIH Funding of COVID Research in 2020: A Preliminary Report.” Johns Hopkins University. August 2021. Accessed January 13, 2022. https://a2e0dcdc-3168-4345-9e39-788b0a5bb779.filesusr.com/ugd/29ca8c_81c3ca04ec5647b49421005934bfbabb.pdf. ↩︎
-
Collison, Patrick; Hsu, Patrick; Cowen, Tyler. “What We Learned Doing Fast Grants.” Future. June 15, 2021. Accessed February 7, 2022. https://future.a16z.com/what-we-learned-doing-fast-grants/. ↩︎
-
“COIVD-19 Funded Research Projects.” NIH. Accessed January 13, 2022. https://covid19.nih.gov/funding#:~:text=NIH%20has%20received%20almost%20%244.9,coming%20from%20the%20U.S.%20Congress%20. ↩︎
-
I counted Harvard four times since it is affiliated with four of the top 50 institutions. I only counted MIT once even though it also has a stake in the Broad Institute because that overlaps with Harvard’s stake. ↩︎
-
“Agency Profile: National Institutes of Health.” Open Secrets. Accessed January 13, 2022. https://www.opensecrets.org/federal-lobbying/agencies/lobbyists?cycle=2020&id=139. ↩︎
-
“Client Profile: The Association of American Universities.” Open Secrets. Accessed January 13, 2022. https://www.opensecrets.org/federal-lobbying/clients/summary?cycle=2020&id=D000047380. ↩︎
-
“Client Profile: Association of Public & Land-Grant Universities.” Open Secrets. Accessed January 13, 2022. https://www.opensecrets.org/federal-lobbying/clients/summary?cycle=2020&id=D000047380. ↩︎
-
“What We Do.” Research!America. Accessed January 13, 2022. https://www.researchamerica.org/about-us. ↩︎
-
“Research!America Form 990.” Research!America. Accessed January 13, 2022. https://www.researchamerica.org/sites/default/files/Financial_Governance/2020ResearchAmerica990.pdf. ↩︎
-
Note – This includes multiple institutes within the same overlapping organization, for instance, the Ohio State, Ohio State University College of Medicine and the Ohio State University College of Dentistry are all counted separately. ↩︎
-
“Member Organizations.” Research!America. Accessed January 13, 2022. https://www.researchamerica.org/about-us/member-organizations. ↩︎
-
I can’t find Research!America in public lobbying disclosure databases. This means that they probably don’t lobby in the traditional sense, but rather produce polls, advertisements, and other advocacy efforts. ↩︎
-
“Our Members.” United for Medical Research. Accessed January 14, 2022. https://www.unitedformedicalresearch.org/about-umr/. ↩︎
-
“Client Profile: United for Medical Research.” Open Secrets. Accessed January 14, 2022. https://www.opensecrets.org/federal-lobbying/clients/summary?cycle=2020&id=F208632. ↩︎
-
“New Research Finds that Special Interest Lobbying Does Influence NIH Research Funding.” NYU Stern. November 6, 2014. Accessed January 13, 2022. https://www.stern.nyu.edu/experience-stern/news-events/new-research-finds-special-interest-lobbying-does-influence-nih-research-funding. ↩︎
-
Selfridge, Maria. “The 10 largest private-sector employers in Greater Baltimore.” Baltimore Business Journal. July 26, 2019. Accessed February 11, 2022. https://www.bizjournals.com/baltimore/news/2019/07/26/the-10-largest-private-sector-employers-in-greater.html. ↩︎
-
“Largest Employers in Los Angele County.” Los Angeles Almanac. Accessed February 11, 2022. http://www.laalmanac.com/employment/em21e.php. ↩︎
-
“Careers.” Fred Hutchinson Cancer Research Center. Fred Hutch. Accessed February 11, 2022. https://www.fredhutch.org/en/about.html. ↩︎
-
“About Us.” Research Triangle Institute. Accessed February 11, 2022. https://www.rti.org/about-us. ↩︎
-
Varmus, Harold. “Chapter 11 Priority Setting.” NCBI. 2007. Accessed January 14, 2022. https://www.ncbi.nlm.nih.gov/books/NBK190605/. ↩︎